Chemistry: Difference between revisions
(→Elements: adding periodic table image) |
|||
| Line 63: | Line 63: | ||
=== Elements === | === Elements === | ||
{{main|Chemical elements|Periodic table of elements}} | {{main|Chemical elements|Periodic table of elements}} | ||
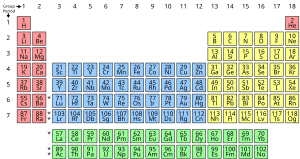
An ''element'' is a class of atoms that have the same number of protons in the nucleus. This number is known as the [[atomic number]] of the element. For example, all atoms with 6 protons in their nuclei are atoms of the element [[carbon]], and all atoms with 92 protons in their nuclei are atoms of [[uranium]]. The most convenient presentation of the chemical elements is in the [[Periodic table of elements|periodic table]] of the chemical elements, which groups elements by atomic number. Due to its ingenious arrangement, | An ''element'' is a class of atoms that have the same number of protons in the nucleus. This number is known as the [[atomic number]] of the element. For example, all atoms with 6 protons in their nuclei are atoms of the element [[carbon]], and all atoms with 92 protons in their nuclei are atoms of [[uranium]]. The most convenient presentation of the chemical elements is in the [[Periodic table of elements|periodic table]] of the chemical elements, which groups elements by atomic number. Due to its ingenious arrangement, periodic table groups (columns) and periods (rows) of elements in the table either share several chemical properties or follow a certain trend in characteristics such as [[atomic radius]], electronegativity, and electron affinity. Lists of the elements by name, symbol, and atomic number are also available. In addition, several [[isotope]]s of an element may exist. | ||
[[File:Simple Periodic Table Chart-blocks.svg|thumb|alt=The periodic table of the elements|Periodic table of the elements]] | |||
=== Ions === | === Ions === | ||
Revision as of 09:52, 29 July 2024
Chemistry is the science of materials. Chemists consider that all of the materials in the world are matter, primarily made up of atoms. The combination of at least two atoms connected by a chemical bond leads to molecules. Ions are derived from atoms or molecules by loss or gain of one or more electrons leading to charged particles. Salts are composed of cations (positively charged ions) and anions (negatively charged ions), so that the substance or material is neutrally charged (without net charge). Chemists use their view of matter at the atomic to molecular level to explain how different materials interact, and how they change under varying conditions. Chemists can induce practical changes of substances, and make new compounds including drugs, explosives, cosmetics, and foods. Chemical synthesis of materials involves bringing together substances in bulk under conditions that they can interact to give different substances. These interactions are called chemical reactions, and always involve rearrangement of electrons around the reacting atoms of each molecule.
The formation of bonds between atoms or molecules means sharing electrons between the composite atoms, and this can transform one substance into another; such as the synthesis of water (H2O) from two gases: hydrogen (H2) and oxygen (O2). Some chemical interactions require energy: the substances must be mixed and heated for a chemical reaction to occur. These energy requiring reactions are called endothermic reactions. Whereas other chemical reactions release heat, and those energy releasing reactions are called exothermic reactions. On a molecular level, reactions can also be initiated by the addition or removal of electrons using electromagnetic radiation (light).
Introduction
Chemistry is about the electrical or electrostatical interactions of matter. These interactions might be between two substances, or between matter (electrons) and energy, especially in conjunction with the First Law of Thermodynamics. Traditionally, chemistry involves interactions between the electrons of substances in chemical reactions, where one or more substances are changed into other substances. Sometimes these reactions are facilitated and enhanced in efficiency by a catalyst, which might be another chemical substance (such as sulfuric acid catalyzing the electrolysis of water), or a non-material phenomenon (such as electromagnetic radiation in photochemical reactions). Traditional chemistry also deals with the analysis of chemicals both in and apart from a reaction, as in spectroscopy.
Ordinary matter consists of atoms and the subatomic components that make up atoms: protons, electrons and neutrons. Atoms can be combined to produce more complex forms of matter such as ions, molecules, or crystals. The structure of the world we experience, and the properties of the matter we interact with, are determined by the properties of chemical substances and their interactions. Steel is harder than iron due to the incorporation of carbon resulting in a more rigid crystalline lattice. Wood burns or undergoes rapid oxidation because it can react spontaneously with oxygen in a chemical reaction above a certain temperature.
Substances tend to be classified in terms of their energy or phase as well as their chemical compositions. The three phases of matter are solid, liquid, and gas. Solids at room temperature have low kinetic energy and are fixed structures which, due to their tight bonds, can resist gravity and other weak forces attempting to rearrange them. Liquids have weaker bonds, with no structure, and they flow with gravity. Gases have no bonds and act as free particles. Water (H2O) is a liquid at room temperature because its molecules are bound by intermolecular forces called hydrogen bonds. Hydrogen sulphide (H2S) on the other hand is a gas at room temperature and pressure, as its molecules are bound by weaker hydrogen bonds dipole-dipole interactions. Due to the higher electronegativity of oxygen compared to that of sulphur, the individual atoms in water tend to have higher charges than in hydrogen sulphide, leading to stronger hydrogen bonding in H2O than in H2S. Hydrogen bonds in water have enough energy to keep the water molecules from separating from each other but not enough to stop them from 'sliding around', making it a liquid at temperatures between 0 and 100° C at sea level. Lowering the temperature or energy allows tighter organized bonds to form, creating a solid, and releasing energy. In this, water (H2O) behaves anomalously, as its greatest density as a fluid is at 4˚ C, not at its freezing point. Increasing the energy heat of fusion will melt the ice, although the temperature will not change until all the ice is melted. Increasing the temperature of the water will cause boiling (see heat of vaporization) when there is enough energy to break the weak polar bonds at 100 °C, allowing the H2O molecules to disperse enough to be a gas. Note that in each case, energy is required to break the hydrogen bonds, as well as to further separate the molecules from each other.
History of chemistry
The roots of chemistry can be traced to the phenomenon of burning. Fire was a mystical force that transformed one substance into another and was of primary interest to mankind. It was fire that led to the discovery of iron and glass. After gold was discovered, and became used as a precious metal, many people were interested to find a method that could convert other substances into gold. This led to the protoscience called alchemy. Alchemists discovered many chemical processes that led to the development of modern chemistry. Chemistry as we know it today was invented by Antoine Lavoisier with his law of Conservation of mass in 1783. The discovery of the chemical elements has a long history, culminating in the creation of the periodic table of the chemical elements by Dmitri Mendeleev. The Nobel Prize in Chemistry, created in 1901, gives an excellent overview of chemical discovery in the past 100 years.
The chemical industry represents an important economic activity. The global top 50 chemical producers in 2004 had sales of 587 billion dollars with a profit margin of 8.1% and research and development spending of 2.1% of total chemical sales.[1]
Subdisciplines of chemistry
Chemistry typically is divided into several major subdisciplines. There are also several main cross-disciplinary and more specialized fields of chemistry.
- Analytical chemistry is the analysis of material samples to gain an understanding of their chemical composition and structure. Analytical chemistry incorporates standardized experimental methods in chemistry. These methods may be used in all subdisciplines of chemistry, excluding purely theoretical chemistry.
- Biochemistry is the study of the chemicals, chemical reactions and interactions in living or semiliving (viruses) organisms. Biochemistry and organic chemistry are closely related, as in medicinal chemistry, radiochemistry, or neurochemistry. Biochemistry is also associated with molecular biology and genetics.
- Inorganic chemistry is the study of the properties and reactions of inorganic compounds. The distinction between organic and inorganic disciplines is not absolute and there is much overlap, most importantly in the subdiscipline of organometallic chemistry.
- Organic chemistry is the study of the preparation and characterization of organic carbon compounds. The number of known organic compounds ranges in the millions, owing to carbon's distinctive ability to form long chains and rings while simultaneously bonding with hydrogen or peripheral functional groups. Organic chemistry is of great commercial importance, providing the underpinnings for the lucrative pharmaceutical and synthetic polymer industries.
- Physical chemistry is the study of the physical and fundamental basis of chemical systems and processes. In particular, the energetics and dynamics of such systems and processes are of interest to physical chemists. Important areas include chemical thermodynamics, chemical kinetics, electrochemistry, statistical thermodynamics, statistical mechanics, spectroscopy, macromolecular chemistry, and magnetic resonance. Physical chemistry largely overlaps with molecular physics, and involves the use of calculus and mathematics in deriving equations. It is usually associated with quantum chemistry, theoretical chemistry, and computational chemistry.
- Polymer chemistry (or macromolecular chemistry) is the study of the chemistry of polymers. Polymer chemistry is considered an area of specialty within the larger field of polymer science, a cross-disciplinary endeavor that encompasses a broad spectrum of researchers. Though firmly rooted in organic synthesis and physical organic chemistry, the work of polymer chemists may overlap with many other disciplines, especially materials science, biology and biochemistry, polymer physics, and medicine.
- Theoretical chemistry is the study of chemistry by fundamental theoretical reasoning (usually within mathematics or physics). In particular, the application of quantum mechanics to chemistry is called quantum chemistry. Since the end of the Second World War, the development of computers has allowed a systematic development of computational chemistry, which is the art of developing and applying computer programs for solving chemical problems. Theoretical chemistry overlaps with (theoretical and experimental) condensed matter physics and molecular physics.
- Nuclear chemistry is the study of the chemistry of radioactive materials, the chemical effects of radiation and all chemistry associated with nuclear equipment and processes. Modern transmutation is a large component of nuclear chemistry, and the table of nuclides is an important result and tool for this field.
Other areas of specialty within chemistry include astrochemistry, atmospheric chemistry, chemo-informatics, electrochemistry, environmental chemistry, flow chemistry or rheology, geochemistry, green chemistry, medicinal chemistry, organometallic chemistry, petrochemistry, pharmacology, photochemistry, phytochemistry, solid-state chemistry, sonochemistry, supramolecular chemistry, surface chemistry, and thermochemistry.
Other technical fields with strong ties to chemistry include chemical engineering, materials science, medicine, metallurgy, molecular biology, molecular genetics, nanoscience or nanotechnology, pharmacology and many subdisciplines within the field of physics. Social sciences with ties to chemistry include the study of science, technology, and society, and the history of science.
Fundamental concepts
Nomenclature
Nomenclature refers to the system for naming chemical compounds. There are well-defined systems in place for naming chemical species. IUPAC nomenclature describes the complete structural information of small (monomeric) molecules. Organic compounds are named according to the organic nomenclature system. Inorganic compounds are named according to the inorganic nomenclature system.
"Common names," or chemical names not based on the IUPAC nomenclature system, are frequently used for many substances. These names may be preferable as the IUPAC name may be very long (over 50 characters). Materials Safety Data Sheets require the listing of all common names as well as IUPAC names for a given substance.
Atoms
An atom is a collection of matter consisting of a positively charged core (the atomic nucleus), which contains protons and neutrons, and which maintains a number of electrons to balance the positive charge in the nucleus.
Elements
An element is a class of atoms that have the same number of protons in the nucleus. This number is known as the atomic number of the element. For example, all atoms with 6 protons in their nuclei are atoms of the element carbon, and all atoms with 92 protons in their nuclei are atoms of uranium. The most convenient presentation of the chemical elements is in the periodic table of the chemical elements, which groups elements by atomic number. Due to its ingenious arrangement, periodic table groups (columns) and periods (rows) of elements in the table either share several chemical properties or follow a certain trend in characteristics such as atomic radius, electronegativity, and electron affinity. Lists of the elements by name, symbol, and atomic number are also available. In addition, several isotopes of an element may exist.
Ions
An ion is a charged species, or an atom or a molecule that has lost or gained one or more electrons. Positively charged cations (e.g., sodium cation Na+) and negatively charged anions (e.g., chloride Cl–) can form neutral salts (e.g., sodium chloride NaCl). Examples of polyatomic ions that do not split up during acid-base reactions are hydroxide (OH–) and phosphate (PO43–).
Compounds
A compound is a substance with a fixed ratio of chemical elements, which determines the composition, and a particular organization, which determines chemical properties. For example, water is a compound containing hydrogen and oxygen atoms in the ratio of two to one, with the oxygen atom forming a chemical bond with each of the two hydrogen atoms and the three atoms forming an angle of 104.5°. Compounds are formed and interconverted by chemical reactions.
Molecules
A molecule is a combination of two or more atoms in a definite arrangement held together by chemical bonds. It is the smallest indivisible portion of a pure compound or element that retains a set of unique chemical properties.
Substance
A chemical substance can be an element, compound or a mixture of compounds, elements or compounds and elements. Most of the matter we encounter in our daily life are one or another kind of mixtures, e.g. air, alloys, biomass etc.
Bonding
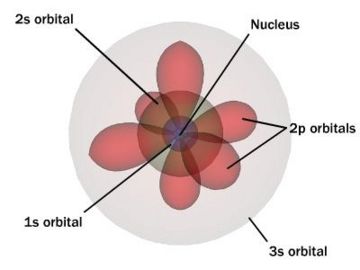
A chemical bond is the multipole balance between the positive charges in the nuclei and the negative charges oscillating about them. More than simple attraction and repulsion, the energies and distributions characterize the availability of an electron to bond to another atom. These potentials create the interactions that hold atoms together in molecules or crystals. In many simple compounds, Valence Bond Theory, the Valence Shell Electron Pair Repulsion model (VSEPR), and the concept of oxidation number can be used to predict molecular structure and composition. Similarly, theories from classical physics can be used to predict many ionic structures. With more complicated compounds, such as metal complexes, valence bond theory fails and alternative approaches, primarily based on principles of quantum chemistry such as the molecular orbital theory, are necessary. See diagram on electronic orbitals.
States of matter
A phase is a set of states of a chemical system that have similar bulk structural properties, over a range of conditions, such as pressure or temperature. Physical properties, such as density and refractive index tend to fall within values characteristic of the phase. The phase of matter is defined by the phase transition, which is when energy put into or taken out of the system goes into rearranging the structure of the system, instead of changing the bulk conditions.
Sometimes the distinction between phases can be continuous instead of having a discrete boundary, in this case the matter is considered to be in a supercritical state. When three states meet based on the conditions, it is known as a triple point and since this is invariant, it is a convenient way to define a set of conditions.
The most familiar examples of phases are solids, liquids, and gases. Less familiar phases include plasmas, Bose-Einstein condensates and fermionic condensates and the paramagnetic and ferromagnetic phases of magnetic materials. Even the familiar ice has many different phases, depending on the pressure and temperature of the system. While most familiar phases deal with three-dimensional systems, it is also possible to define analogs in two-dimensional systems, which has received attention for its relevance to systems in biology.
Chemical reactions
A Chemical reaction is a process that results in the interconversion of chemical substances. Such reactions can result in molecules attaching to each other to form larger molecules, molecules breaking apart to form two or more smaller molecules, or rearrangement of atoms within or across molecules. Chemical reactions usually involve the making or breaking of chemical bonds. For example, substances that react with oxygen to produce other substances are said to undergo oxidation; similarly a group of substances called acids or alkalis can react with one another to neutralize each other's effect, a phenomenon known as neutralization. Substances can also be dissociated or synthesized from other substances by various different chemical processes.
Quantum chemistry
Quantum chemistry mathematically describes the fundamental behavior of matter at the molecular scale. It is, in principle, possible to describe all chemical systems using this theory. In practice, only the most simple chemical systems may qualitatively be investigated in purely quantum mechanical terms, and approximations must be made for most practical purposes (e.g., Hartree-Fock, post Hartree-Fock or density functional theory, see computational chemistry for more details). Hence a detailed understanding of quantum mechanics is not primarily necessary for most chemistry, as the important implications of the theory (principally the orbital approximation) can be understood and applied in simpler terms.
In quantum mechanics (several applications in computational chemistry and quantum chemistry), the Hamiltonian, or the physical state, of a particle can be expressed as the sum of two operators, one corresponding to kinetic energy and the other to potential energy. The Hamiltonian of a particle with no electric charge and no spin is described by the Schrödinger wave equation of a particle without forces being exerted on it.
Solutions of the Schrödinger equation for the hydrogen atom gives the form of the wave function for atomic orbitals, and the relative energy of say the 1s,2s,2p and 3s orbitals. The orbital approximation can be extended to understand the other atoms e.g. helium, lithium and carbon.
Chemical laws
The most fundamental concept in chemistry is the law of conservation of mass, which states that there is no detectable change in the quantity of matter during an ordinary chemical reaction. Modern physics shows that it is actually energy that is conserved, and that mass is simply one of several forms of energy, a concept that becomes important in nuclear chemistry. Conservation of energy leads to the important concepts of equilibrium, thermodynamics, and kinetics.
Further laws of chemistry elaborate on the law of conservation of mass. Joseph Proust's law of definite composition says that pure chemicals are composed of elements in a definite formulation; we now know that the structural arrangement of these elements is also important.
Dalton's law of multiple proportions says that these chemicals will present themselves in proportions that are small whole numbers (i.e. 1:2 O:H in water), although in many systems (notably biomacromolecules and minerals) the ratios tend to require large numbers, and are frequently represented as a fraction. Such compounds are known as non-stoichiometric compounds.
Etymology
- Main article: Etymology of alchemy
The word chemistry comes from the earlier study of alchemy, from the old French alkemie; and the Arabic al-kimia: "the art of transformation." An alchemist was called a 'chemist' in popular speech, and later the suffix "-ry" was added to this to describe the art of the chemist as "chemistry".
Alchemy in turn is thought to possibly derive either from Greek word chemeia (χημεία) meaning "cast together", "pour together", "weld", "alloy", etc. (from khumatos, "that which is poured out, an ingot"), or from the Coptic name for Egypt kēme, or alternately, from Persian kimia meaning "gold".
References
- ↑ (July 18, 2005) "Top 50 Chemical Producers". Chemical & Engineering News 83 (29): 20–23.