Tuberculosis
Tuberculosis, ("TB" or "consumption") is a disease caused by the presence of the organism Mycobacterium tuberculosis in the tissues of the body. "If properly treated, tuberculosis caused by drug-susceptible strains is curable in virtually all cases. If untreated, the disease may be fatal within 5 years in more than half of cases."[1] "The World Health Organization (WHO) estimates that 1722 million are infected with Mycobacterium tuberculosis." [2] Although tuberculosis is a lung disease in most people, it affects other organs in at least a third of patients.[3]
There are several other species of the genus Mycobacterium that are also human pathogens, but all of these organisms are of much lower virulence than Mycobacterium tuberculosis. That means that they are less apt to be able to invade the body and cause illness, and that even when they succeed in infecting a person, they cause less serious infection that is more often managed successfully by the body's own immune defenses. When such "minor" species of Mycobacteria cause disease in humans the illness is called atypical tuberculosis, not tuberculosis.
This article is restricted to tuberculosis infection by Mycobacterium tuberculosis. Despite that narrow focus, the manifestations of human infection by this one organism are so variable that the article can only briefly discuss the more common presentations. Infections by M. tuberculosis range from a walled-off primary nidus of infection that causes no symptoms of disease (latent TB) to localized illness of the lungs, lymph nodes, or other body parts like the ears, voice box, that can range from mild to disabling, to a disseminated infection that is seeded throughout the entire body (Miliary TB) and is frequently fatal even with the most aggressive of available treatments.
As with organisms in any species, including bacteria, there are variations from one individual to the next. The tubercle bacillus is well known for individual strains that vary, independently, in both virulence and antibiotic resistance. In general, the severity of disease caused by TB infection is dependent on three major factors- the virulence (and dose) of the particular strain of TB a patient was exposed to, (2) the patients own physical integrity and immune competency (host resistance), and (3) timely treatment (or lack of timely treatment) with an appropriate selection of anti-tuberculosis drugs for a sufficiently prolonged period.
Epidemiology
Approximately one-third of the world population is infected with TB at any given time, with approximately 2 million TB-related deaths per year worldwide.
People with HIV disease, cancer, malnutrition, chemotherapy, drug or alcohol abuse are especially susceptible to developing active TB. TB rates and deaths are greatest in the developing world, and TB is one of the leading causes of death in people with HIV. In the United States, TB rates increased during the early years of the HIV epidemic, but have since declined, and are in fact the lowest they have been since surveillance began in 1953. However, TB still affects tens of thousands of Americans, and kills several hundred per year.
Globally, the highest rates of tuberculosis infection are in , and additional geographic regions with high endemic rates of infection include .
Infection in other species
Mycobacterium tuberculosis affects other vertebrates besides humans, and can be transmitted between species, however other species of Mycobacterium are more commonly contracted by non-human vertebrates. "Tuberculosis in cattle has been known even before classical times... Owing to its close resemblance with the disease in humans and the close relationships between the causative agents, they were initially termed M. tuberculosis typus bovinus and M. tuberculosis typus humanus."[4]
Elephants have been diagnosed with the disease, in captivity.[5]
One of the risks of non-pasteurized milk is drinking infected secretions from cows afflicted with bovine tuberculosis. Efforts by public health officials to inspect cattle were, however, resisted by dairy farmers in the early 20th century.[6]
Microbiology
Natural History
Although it is possible to live with this infection for years, untreated, in the era "before effective drugs were available, 50% of patients with active pulmonary tuberculosis died within 2 years, and only 25% were cured." From: Chapter 248 - Mycobacterium tuberculosis TREATMENT OF TUBERCULOSIS in Mandell, Bennett, & Dolin: Principles and Practice of Infectious Diseases, 6th ed.
Tuberculosis can affect any organ in the body, but the primary disease is in the lungs. TB is usually spread by prolonged contact with someone with active pulmonary TB. This most often occurs in household contacts, or in people who work with an infected person in a workplace with poor ventilation.
Pathology
When tuberculosis infects tissue the hallmark lesion is a granuloma. Histologically, the granuloma may contain Langhans giant cells which are multinucleated giant cells whose nuclei are situated peripherally within in cell in a horseshoe-shaped pattern.[7][8]Langhans giant cells are named for Theodor Langhans.[9]
Latent Tuberculosis
In most people, especially those who have a normal immune system, after becoming infected, the body controls the illness. They have no symptoms, and are not contagious. Signs of infection can be a positive TB test or typical changes on X-rays of the chest. In a small, but significant, percentage of people, latent TB can "reactivate" and make the person ill. Therefore, most people with latent TB are treated to eliminate the infection.
Active Pulmonary Tuberculosis
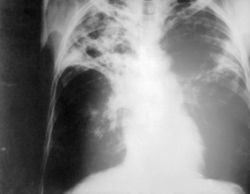
In some people, the initial infection can make them ill, in others, latent TB reactivates and makes them ill. In pulmonary tuberculosis the usual symptoms are cough for longer than three weeks, bloody sputum, fevers, night sweats, and weight loss. At this stage, the disease is very contagious, and is likely to eventually be fatal if not treated.
Extrapulmonary Tuberculosis
Gastrointestinal tuberculosis
When people with pulmonary tuberculosis are not treated with an effective combination of antibiotics, the bacteria multiply and are present, in great and ever increasing quantity, in the secretions coughed up from the lungs. This sputum touches the lining of the mouth, the back of the nose, and is also swallowed, and, unless treatment to stop the multiplication of the bacteria is instituted, lodges in the stomach and intestines of patients with pulmonary TB. Even though stomach acid has some killing effect on the bacteria, if the numbers are large enough, there are survivors that exit the anus. At any point along the uper airway tract and gastrointestinal tract, from the lips, the nostrils, all the way to the anus, adherence of the bacteria may lead to an infection, with the formation of a non-healing ulcer.
About 70% of individuals with advanced stages of pulmonary tuberculosis have active infections in their GI tract seeded through swallowing secretions coughed up from the lungs, and so have both pulmonary TB and a form of extrapulmonary TB that was contracted as a direct result of the pulmonary infection.
Laryngeal Tuberculosis
Osteomyelitis
Pott's Disease is osteomyelitis of the vertebrae.[10]
Tuberculous osteomyelitis of the mandible can mimic a dento-alveolar abscess.[11]
Tuberculosis Adenitis ("Scrofula")
Scrofula is tuberculous infection of the cervical lymph nodes.[12]
Disseminated Tuberculosis (Miliary TB)
Diagnosis
A clinical prediction rule can estimate which patients with pneumonia might have tuberculosis.[14] Patients without any of the following findings are low risk:
- Cavitatary lesion on chest radiograph
- Apical infiltrate on chest radiograph
- Immigrant
- Weight loss
- Positive TB history (includes previous positive tuberculin skin test result)
- Homeless
- Incarcerated
Because of difficulties with the Tuberculin skin test, many laboratory methods of diagnosis are emerging. These tests have been reviewed in detail.[15][16]
Adenosine deaminase
In 2007, a systematic review of adenosine deaminase by the NHS Health Technology Assessment Programme concluded "There is no evidence to support the use of ADA tests for the diagnosis of pulmonary TB. However, there is considerable evidence to support their use in pleural fluid samples for diagnosis of pleural TB, where sensitivity was very high, and to a slightly lesser extent for TB meningitis. In both pleural TB and TB meningitis, ADA tests had higher sensitivity than any other tests."[16]
Nucleic acid amplification tests (NAAT)
This is a heterogeneous group of tests that use polymerase chain reaction (PCR) to detect mycobacterial nucleic acid. These test vary in which nucleic acid sequence they detect and vary in their accuracy. The two most common commercially available tests are the amplified mycobacterium tuberculosis direct test (MTD, Gen-Probe) and Amplicor (Roche Diagnostics). In 2007, a systematic review of NAAT by the NHS Health Technology Assessment Programme concluded that "NAAT test accuracy to be far superior when applied to respiratory samples as opposed to other specimens. Although the results were not statistically significant, the AMTD test appears to perform better than other currently available commercial tests."[16]
Interferon-γ release assays
In 2007, two systematic reviews of Interferon-γ (interferon-gamma) release assays (IGRAs) concluded the tests noted excellent specificity and the ability of the tests to distinguish latent TB from prior vaccination.[17][16]
The ELISpotPLUS assay is more sensitive than the original ELISpot assay. Patients who are negative on both skin testing and the ELISpotPLUS assay are very unlikely to have tuberculosis.[18] However, this test has not been well studied in immunosuppressed patients.
Treatment
"A fundamental problem in the treatment of tuberculosis (TB) is the long duration of therapy required for cure."[19] Although anti-bacterial drugs given to treat other infections are rarely if ever called "chemotherapy", that term is ordinarily applied to the courses of antibiotics used for the treatment of Mycobacterium tuberculosis. Despite the use of the word, none of these medications are similar to the anti-cancer drugs that are the agents most often meant by the term.
In all bacteria infections, antibiotic treatment affects the bacteria when they are actively reproducing. That means that bacteria like M. tuberculosis are not quick to be killed even by antibiotics that are capable of killing them, because the bacteria can become dormant and reproduce very slowly. Additionally, the tubercle bacillus can be protected by the tissue damage that it causes. In other words, when tuberculosis causes pulmonary tissue and lymph nodes to become necrotic, the blood supply to these tissues is compromised and even if the patient is taking an adequate dose of a set of antibiotics that will kill replicating bacteria, those bacteria in the tissues that do not receive a normal blood supply will not be exposed to the antibiotics in the blood.
Additionally, the patient's immune system is also designed to kill the bacteria when they are dividing and is less able to eradicate a dormant infection than the same number of bacteria that are actively dividing. Further, the necrotic and cavitary tissue sequesters the bacteria from the circulating cells and antibodies of the immune system, and protects the bacteria from the patient's host defenses.
If the bacteria are exposed to antibiotics that do not wipe them out, then resistant organisms may multiply and, through genetic factors, drug resistant strains can become the predominant ones in the infection.
Drugs against TB: Antituberculous chemotherapy for treatment, Antituberculous prophylaxis for prevention
The first efficacious drugs to treat TB became available in different parts of the world in the 1940's and 1950's, before that time - treatment consisted on long-term hospitalization that focused both on quarantining the patient from society and on increasing host resistance.
Isoniazid
Rifampin
Pyrazinamide
Ethambutol
Streptomycin
Fluoroquinolones
Latent TB is usually treated fairly easily with one to two medications, depending on local resistance patterns. The treatment often lasts up to a year.
Inpatient and outpatient therapy: hospital or ambulatory care?
Compliance, noncompliance and resistant TB
Multi Drug Resistant TB (MDR-TB): Certain geographical areas have seen rises in "MDR-TB". This is especially a problem when patients are intermittently-compliant with their therapy, as the organism develops drug-resistance. Active TB usually requires hospitalization and treatment with multiple medications. Given that the treatment regimen is lengthy and complicated, Directly Observed Therapy (DOT) is often recommended. There have been many legal and ethical issues involving involuntary isolation of non-compliant patients with active TB.
Directly Observed Therapy (DOT)
Therapy aimed at increasing host resistance
The sanatoriums that served as treatment centers for tuberculosis patients in the pre-antibiotic era helped reduce the spread of infection by effecting a partial quarantine, but also served to increase patient's resistant to the progression of disease by XXX.
Nutrition
Reduction of bacterial load
Decreased host resistance
Immunosuppression
AIDS (HIV)
Organ transplant recipients
Infection control
Public health
Tuberculosis is a priority for the World Health Organization. It has been assessed as a world threat in an U.S. National Intelligence Estimate.
In hospital
Reducing the rate of resistant TB
Historical
Bernier (2005) shows the incidence of pulmonary tuberculosis took on alarming proportions in 18th-century Europe. He examines 12 treatises by learned medical practitioners on the causes of the malady in Great Britain and France. Four etiological models appear to have dominated medical thinking in this context: contagion, physiological disorders, hereditary predispositions, and links to behavior and lifestyles. The hereditary model, which eventually triumphed over the others and became dominant for the better part of the 19th century, can be interpreted within the context of the growth of liberal and individualist ideology.
Tuberculosis was the leading cause of death in the United States in 1900. By the 1920s mortality rates in the general population were decreasing significantly, a decline that had begun around 1850.
Jones (2003)[23] evaluates the effects of tuberculosis on Hong Kong's public housing and public health policies in the first half of the 20th century. Tuberculosis and pneumonia were the biggest causes of death in 1900-40. However, death rates alone fail to reflect the large number of TB sufferers who did not die. The British medical directors in Hong Kong repeatedly deplored the unsanitary, crowded housing situation in Hong Kong, as well as the Chinese habit of public spitting, as factors in in spreading disease. From 1911, an anti-spitting educational campaign was pursued but with little real success. Hong Kong also suffered from a lack of sanitoriums and hospital space to isolate TB patients. Public housing commissions had estimable proposals during this period but could not find financing as the international situation deteriorated and Hong Kong was seized by the Japanese in 1941. Subsidized housing did not occur until after World War II.
Abel (2003)[24] finds the identification of Mexicans with tuberculosis by public health officials in Los Angeles helped to drive policies of exclusion in immigration and health care restrictions and, later, expulsion in the forms of deportation and repatriation. After 1914, public officials tried to prevent all poor consumptives from taking up residence within California. During the 1920's, two reports by the California State Board of Health argued that an influx of sick Mexican workers endangered public health and placed an insupportable economic burden on the state. Although harsh living and working conditions in Los Angeles may have caused a disproportionately high rate of incidence of tuberculosis among Mexican immigrants, contemporary statistics were probably inflated, reflecting the preconception that Mexicans were particularly susceptible to the disease. During the Great Depression, such arguments helped to justify large-scale deportation and repatriation campaigns.
Native Americans
Southerton (2004)[25] examines the efforts of Indian Service physician James R. Walker to reduce the spread of tuberculosis on the Pine Ridge Indian Reservation in South Dakota between 1896 and 1914. After Walker concluded that the greatly increased prevalence of tuberculosis among Lakota at Pine Ridge was due to their change from a nomadic lifestyle to one of cabin residency, his education efforts dramatically reduced the number of cases. Walker also sought to create a tent camp sanatorium, arguing that it would be well-ventilated, easily sanitized, and cost effective. Walker's proposal was rejected, partly because his interest in Indian medical rituals was believed to encourage "heathenish" practices among natives.
MacMahon (2003)[26] explores the experience of tuberculosis among the Navajos between 1920 and 1960. There was ineffective communication between the Navajos and Western healthcare providers and a serious underestimation by Federal officials of the importance of traditional culture in Dine lives. The 1930s livestock reduction ordered by Commissioner of Indian Affairs John Collier contributed significantly to the mental anguish and malnutrition among the population and had a negative impact on the tribe's subsistence economy. The poverty ensuing from herd reduction resulted in an increased susceptibility to tuberculosis and other diseases. These events occurred in an era when tuberculosis morbidity rates in New Mexico and Arizona were the highest in the country and when the Navajos, who had not been disease-free, were experiencing increased contact with off-reservation inhabitants. Crowding, wretched diets, and inadequate healthcare at boarding schools also led many Navajo children to contract the disease. Physicians sent children with tuberculosis home, which spread the infection to their families. During the 1930s, communications improved between the Navajos and more culturally sensitive physicians, resulting in increased Navajo acceptance of care. However, health care availability and mutual respect declined precipitously during World War II. Progress nearly ceased in tuberculosis efforts. By 1953 tuberculosis was epidemic among the Navajos. Cultural misunderstanding by health professionals, and Federal officials as well, contributed to the longevity of the tuberculosis issues within the tribe. A central tenet of Navajo belief is that everything in life is connected and to be well is to live in harmony. Navajos eagerly sought benefit of other cultures that could meld with their own, allowing them to maintain their traditions even as they utilized Western health care. Indian Service officials believed tuberculosis was a disease that simply required a medical solution. Instead, it demanded social solutions and cultural respect, as well, of the Navajos and the medical community alike.
Historical therapies
Grad (2004) examines the use of cod liver oil as a treatment for pulmonary tuberculosis between the 1770s and the 1930s, tracing attempts to understand the disease from the work of American physician Benjamin Rush onward. Cod liver oil was introduced into the British pharmacopoeia in 1771, and by the end of the 18th century it was a prominent treatment in the Manchester Infirmary. In 1841 Edinburgh physician John Hughes Bennett (1812-75) published a treatise on cod liver oil, detailing its use on tuberculosis. Bennett's successes led other physicians in Britain to investigate the effects of cod liver oil on tuberculosis. Cod liver oil entered medical practice in the United States in the 1840s, quickly gaining widespread use. After the discovery of the tubercle bacillus, some claimed that cod liver oil had specific action against the bacillus. In the early 20th century, after the discovery that cod liver oil contained Vitamin D, its therapeutic use was focused on the treatment of rickets.
Benedek (2004) examines the history of the treatment of tuberculosis with gold therapy. Despite a wealth of negative experimental evidence, gold therapy was popular in the early to mid-20th century. Gold therapy first appeared in the work of Robert Koch (1843-1910), who found gold cyanide to be effective against cultures of tuberculosis, though not in animals infected with the bacteria. The 1920's saw a rise in the popularity of gold therapy, largely through the work of Danish physicians Holger Mollgaand, Knud Faber, and Knud Secher, who reintroduced Koch's methods despite their earlier nonsuccess. By the mid-1920's, the therapy had become widespread and continued to be popular in Europe and the United States, even as it failed clinical trials, well into the 1940's.
Bonah (2005) shows the anti-tuberculosis Bacillus Calmette-Guérin (BCG) vaccine was conceived and developed between 1905 and 1921 at the Pasteur Institute in France. Between 1921 and Albert Calmette's death in 1933, the vaccine went through a first period of national and international production and distribution for use in humans. In France these activities were exclusively carried out by Calmette and his collaborators at the Pasteur Institute in Paris. Initial improvised production in a small room in the cellar gave way in 1931 to the construction of spacious new laboratories for research on tuberculosis and the preparation of the BCG within the premises of the Pasteur Institute. In France the BCG was conceived of not as a commercial speciality but was to be distributed free of charge. The technical monopoly of its production nevertheless lay with the Pasteur Institute, and standardization of scientific proof of safety, efficacy, and stability was also dominated by the institute. In contrast, the international production and distribution of the vaccine was transferred, free of charge, to trustworthy laboratories outside France. Multiplication of producers and users led to an increased need for standardization, both concerning safety, efficacy, and stability of the vaccine and in its medical uses. Whereas standardization was fairly successful in the interwar period in France, the international efforts remained largely unsuccessful until after World War II, when, under Scandinavian leadership and in the context of mass vaccination programs supported by the World Health Organization (WHO) and the UN International Children's Emergency Fund (UNICEF), international standardization was effectively implemented.
Lanigan and Beck (2005) review the treatment of tuberculosis on Prince Edward Island, Canada, during 1888-1931. Probably 20% of the province's deaths resulted from tuberculosis, making Prince Edward Island's tuberculosis mortality rate the highest in Canada. Despite this, few sources of help existed. The province's first sanatorium did not open until 1915 in Wiltshire; it was established by volunteers without government aid, although the province did agree to operate it once it opened. The hospital was taken over as a military hospital during World War I, but the building was destroyed after the war because neither the provincial government nor the federal government would assume the cost of running it. Public pressure finally led to the construction of a provincial sanatorium in Charlottetown in 1931, the same year a provincial Department of Health was established. Even so, Prince Edward Island's tuberculosis rate remained high for decades.
Between 1956 and the mid-1960's, urban South India served as a global "laboratory" for the study of tuberculosis. Amrith (2004) examines the origins and development of two internationally sponsored projects, in Madras (now Chennai) and Bangalore, established to investigate the medical and social implications of introducing antituberculosis drugs in poor urban communities. Amrith thus provides a case study of the role of biomedical technology in an early international health campaign, examining how changing knowledge and technology recast the perceived relationship between disease and poverty. Amrith also situates the history of tuberculosis control within the broader context of the ideas and politics of "development" in the international arena during the 1950's and 1960's. In particular, Amrith looks at the interaction between a medical discourse involving the individualization of tuberculosis as a health problem and an economic discourse of "cost-effectiveness" in international public health policy.
See also
References
- ↑ for quote: Mario C. Raviglione, Richard J. O'Brien: Chapter 150. Tuberculosis in Harrison's Internal Medicine. Harrison's Online
- ↑ Watters DA. Surgery for tuberculosis before and after human immunodeficiency virus infection: a tropical perspective. British Journal of Surgery. 84(1):8-14, 1997 page 8.
- ↑ Mandell, Douglas and Bennett's Principles and Practice of Infectious Diseases, Churchill Livingstone.
- ↑ Michel AL. Implications of tuberculosis in African wildlife and livestock. [Review] [30 refs] [Journal Article. Review] Annals of the New York Academy of Sciences. 969:251-5, 2002 Oct. UI: 12381600
- ↑ Payeur JB. Jarnagin JL. Marquardt JG. Whipple DL. Mycobacterial isolations in captive elephants in the United States. Annals of the New York Academy of Sciences. 969:256-8, 2002 Oct. UI: 12381601
- ↑ In Los Angeles in 1912 the United Milk Producers Association, opposed inspection on the grounds that it would cause milk prices to rise and that scientists were not united on the question of whether tuberculosis could be transmitted from cattle to humans. Edson's proposed ordinance failed to pass due to the public's poor understanding of germ theory. Jennifer Koslow, "Putting it to a Vote: the Provision of Pure Milk in Progressive Era Los Angeles." Journal of the Gilded Age and Progressive Era 2004 3(2): 111-144. Issn: 1537-7814 Fulltext: in History Cooperative.
- ↑ Anonymous (2024), Langhans giant cells (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Baron, Samuel (1996). “Mycobacterium tuberculosis Complex”, Medical Microbiology. Univ of Texas Medical Branch. DOI:url=http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=giant,Langhans&rid=mmed.section.1811. ISBN 0-9631172-1-1.
- ↑ Pritchard J, Foley P, Wong H (2003). "Langerhans and Langhans: what's misleading in a name?". Lancet 362 (9387): 922. DOI:10.1016/S0140-6736(03)14323-1. PMID 13678997. Research Blogging.
- ↑ Anonymous (2024), Pott's Disease (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Bhatt AP, Jayakrishnan A (2001). "Tuberculous osteomyelitis of the mandible: a case report". Int J Paediatr Dent 11 (4): 304–8. DOI:10.1046/j.1365-263X.2001.00276.x. PMID 11570448. Research Blogging.
- ↑ Anonymous (2024), scrofula (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Long R. Guzman R. Greenberg H. Safneck J. Hershfield E. Tuberculous mycotic aneurysm of the aorta: review of published medical and surgical experience.[see comment]. [Review] [74 refs] [Case Reports. Journal Article. Review] Chest. 115(2):522-31, 1999 Feb. UI: 10027455
- ↑ Moran GJ, Barrett TW, Mower WR, et al. (May 2009). "Decision instrument for the isolation of pneumonia patients with suspected pulmonary tuberculosis admitted through US emergency departments". Ann Emerg Med 53 (5): 625–32. DOI:10.1016/j.annemergmed.2008.07.027. PMID 18760503. Research Blogging.
- ↑ Drobniewski F, Caws M, Gibson A, Young D (2003). "Modern laboratory diagnosis of tuberculosis". Lancet Infect Dis 3 (3): 141-7. PMID 12614730.
- ↑ 16.0 16.1 16.2 16.3 Dinnes J, Deeks J, Kunst H, Gibson A, Cummins E, Waugh N, Drobniewski F, Lalvani A (2007). "A systematic review of rapid diagnostic tests for the detection of tuberculosis infection". Health Technol Assess 11 (3): 1-314. PMID 17266837.
- ↑ Menzies D, Pai M, Comstock G (2007). "Meta-analysis: new tests for the diagnosis of latent tuberculosis infection: areas of uncertainty and recommendations for research". Ann. Intern. Med. 146 (5): 340-54. PMID 17339619. [e]
- ↑ Dosanjh, Davinder P.S. et al. 2008. Improved Diagnostic Evaluation of Suspected Tuberculosis. Ann Intern Med 148, no. 5:325-336.
- ↑ "Why Is Long-Term Therapy Required to Cure Tuberculosis?" Connolly LE, Edelstein PH, Ramakrishnan L PLoS Medicine Vol. 4, No. 3, e120 doi:10.1371/journal.pmed.0040120
- ↑ Watters DA. Surgery for tuberculosis before and after human immunodeficiency virus infection: a tropical perspective. [Review] [94 refs] [Journal Article. Review] British Journal of Surgery. 84(1):8-14, 1997 Jan. UI: 9043439
- ↑ Small PM, Hopewell PC, Singh SP, et al. The epidemiology of tuberculosis in San Francisco: a population-study using conventional and molecular methods. N Engl J Med 1994; 330: 1703-09.
- ↑ Aguado JM. Herrero JA. Gavalda J. Torre-Cisneros J. Blanes M. Rufi G. Moreno A. Gurgui M. Hayek M. Lumbreras C. Cantarell C. Clinical presentation and outcome of tuberculosis in kidney, liver, and heart transplant recipients in Spain. Spanish Transplantation Infection Study Group, GESITRA.[erratum appears in Transplantation 1997 Sep 27;64(6):942]. [Review] [22 refs] [Journal Article. Multicenter Study. Review] Transplantation. 63(9):1278-86, 1997 May 15. UI: 9158022
- ↑ Margaret Jones, "Tuberculosis, Housing and the Colonial State: Hong Kong, 1900-1950." Modern Asian Studies 2003 37(3): 653-682. Issn: 0026-749x
- ↑ Emily K. Abel, "From Exclusion to Expulsion: Mexicans and Tuberculosis Control in Los Angeles, 1914-1940." Bulletin of the History of Medicine 2003 77(4): 823-849. Issn: 0007-5140 Fulltext: in Project Muse
- ↑ Don Southerton, "James R. Walker's Campaign Against Tuberculosis on the Pine Ridge Indian Reservation." South Dakota History 2004 34(2): 107-126. Issn: 0361-8676
- ↑ Sandra Varney MacMahon, "Tuberculosis, the Navajos, and Western Healthcare Providers, 1920-1960." PhD dissertation U. of New Mexico 2003. 357 pp. DAI 2003 64(3): 1044-A. DA3085718