Food reward
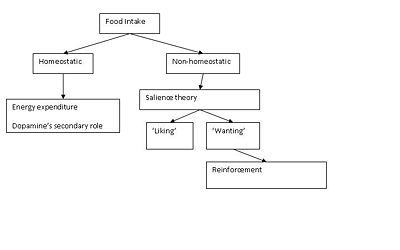
Food intake involves both 'homeostatic feeding' (energy demands) and ‘non-homeostatic feeding’; the latter is associated with food reward, which involves both 'liking’ (pleasure/palatability) and ‘wanting’ (incentive motivation) according to the salience theory. Experiments in mice suggest that ‘liking’ involves the release of mu-opioid peptides in brain, while ‘wanting’ involves the neurotransmitter dopamine [1].
| Key Definitions |
|---|
| Wanting/ Incentive salience - The motivational aspect of a stimulus that transforms the sensory information into a more desirable stimulus. |
| Liking - Immediate pleasure from consumption |
| Hedonia - The feeling of pleasure |
| Motivation- The direction towards a particular behaviour to achieve a goal |
| Reinforcement - The process by which a stimulus strengthens a behavioural response so that the probability of response is increased when the stimulus is presented again. |
Motivated behaviour and food as a reinforcer
The brain’s reward systems react to stimuli such as sight, smell and taste, and other cues that predict food. However, hunger cannot result in unconditioned goal-directed behaviour; [2] chance encounters with palatable foods are required before goal-directed behaviour can occur, which link the internal needs with the salience of environmental stimuli [3]For exa mple, an infant recognises and learns to seek out sweet tastes, but the desire for any particular food is controlled by the interaction of peptide levels (related to hunger) with neural circuits in the brain which store the animal’s past experience of that particular food. [4] Subsequently, the infant will taste both food and non-food objects indiscriminately until it has received reinforcing feedback from enough stimuli. A monkey’s appetite for yellow bananas requires that the monkey learns to relate the sight of the yellow skin of a banana with the sweet taste of the banana, plus the consequences of eating it. Preference for a particular food results only when the post-ingestional consequences of that food ’reinforce’ the tendency to eat that food. For these reasons, food is considered to be a strong reinforcer. When the response of a behaviour stimulated by a reinforcer increases the frequency of that behaviour; that is positive reinforcement or reward learning, and the positive events are called rewards [5]. The reinforcing efficacy of food reward is the ability of the reward to maintain rather than to establish behaviour; consequently the stimulus learning contributes to the response learning.
Food reward pathways
The first evidence for the involvement of dopamine in food reward came from studies in rats, in which dopamine antagonists blocked the rewarding effects of brain stimulation and of psychomotor stimulants[6] [7].
The Mesolimbic Dopaminergic Reward System
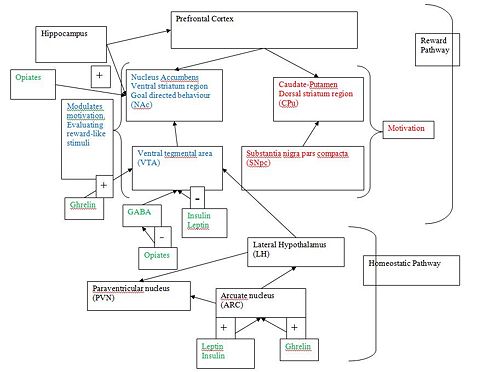
In the ‘reward circuit’, projections from the ventral tegmental area (VTA) to the nucleus accumbens have received the most attention. Dopamine action in the nucleus accumbens is thought to be needed for motivation to acquire food or addictive drugs. Most reviews suggest that the projections from the VTA to the nucleus accumbens are needed for the motivation to eat, but not for food consumption itself.
The Dopamine Hypothesis
Dopamine signalling from the VTA to the nucleus accumbens, hippocampus, amygdala and/or pre-frontal cortex promotes reward-related activities. Dopamine signalling in these brain regions focuses attention to salient environmental events and facilitates behaviour towards directed goals. It is thought that dopamine released from the VTA also promotes learning between food reward and the environment.
Dopamine's role in reward?
•Hedonia – Dopamine in the nucleus accumbens has been said to act as a "pleasure" neurotransmitter. Not all rewards activate the reward system, suggesting that the mesolimbic pathway is not solely hedonic.
•Learning – predictions of future rewards, nucleus accumbens and VTA lesions do not affect this part but lack the motivation for the reward. •Incentive Salience – the ‘wanting’ of the reward, released when there is a stimulus worth working hard for. In absence of dopamine, the environmental stimulus goes unnoticed and the animal will eventually die from starvation and dehydration.
The incentive salience theory seems to best fit the data in this field. Therefore dopamine causes the wanting of the reward after the appropriate stimuli have been processed in the reward system. Dopamine transmission is needed to form these associations. An increase in extracellular dopamine is seen in regard to natural rewards, food, water and sex, during acute administration. However novelty is an important factor in the increased release from the nucleus accumbens. It has been suggested that the role of dopamine in motivation is shared between the nucleus accumbens and CPu pathways(see diagram). The SNpc-CPu pathway is essential for motivation, while dopamine signalling from the VTA to the nucleus accumbens needed in regard to modulating the actions of the other dopamine pathway.
The substantia nigra pars compacta to the caudate putamen
Dopamine-deficient mice starve, apparently because they are not motivated to respond to hunger signals. Thus, it has been proposed that dopamine is crucial for mice to engage in most goal-directed or motivated behaviours. However it is not clear which dopamine pathway is involved; a universal finding is the involvement of the striatum, the input structure of the basal ganglia in a circuit responsible for mediating goal-directed behaviour, with the striatum’s central role being the processing of reward like stimuli. The two proposed pathways are from the VTA to nucleus accumbens (ventral striatum); or the substantia nigra pars compacta to the caudate putamen (dorsal striatum). The striatum includes not only the dorsal region, which encompasses the caudate nucleus and putamen, but also the ventral region that includes the core and shell of the nucleus accumbens[8]
Our understanding of how reward information is processed comes mainly from studies in animal models. One study, using nonhuman primates, found that striatal neurons responded to the anticipation and delivery of reward [9] Another found a reward-related dopamine response specifically in the mouse dorsal striatum, correlated with the delivery of food reward [10] The importance of dopamine in the dorsal striatum was demonstrated using dopamine-deficient mice whose dopamine signalling is restored by viral rescue. These mice learned to lever press for food rewards as quickly as control mice, and their motivation to work for food was restored. Importantly, in these dopamine-deficient mice, feeding was never restored after viral transduction in the nucleus accumbens.[11][12]
Recent advances in neuroimaging has allowed researchers to extend such investigations to the human brain. Dopamine is released in the dorsal striatum of hungry participants when stimulated with food items. During the delivery of rewards, fMRI signals were higher in the dorsal striatum, particularly the head of the caudate putamen(Delgado MR (2007)). It has been proposed that dopamine signaling in the caudate putamen is essential for motivation, while dopamine signaling in the nucleus accumbens modulates this motivation and evaluation of reward-like stimuli.
The effect of hormones on the dopamine reward system
Plasma concentrations of ghrelin, leptin and insulin reflect the size of energy stores, such as adipose tissue, and act at the mediobasal hypothalamus to influence appetite and energy expenditure. These hormones also affect the dopamine reward pathways [13] Receptors for these hormones are located on dopamine neurones in the VTA, and ligand binding results in activation e.g. by ghrelin [14] or inhibition e.g. by insulin or leptin of dopamine signalling to the nucleus accumbens [15]. These alterations in dopamine signalling pathways have complex effects on eating behaviours.
Both insulin and leptin attenuate food reward, reducing the incentive to eat. For example, administering insulin or leptin to rats affected conditional place preference (CPP), which assesses the ability to relate a particular food reward to a particular environment. Rats were fed a high-fat diet and underwent a ‘training period’ before the test involving intracerebroventricular administration of insulin or leptin. CPP was abolished in those rats that received insulin or leptin before or during the test as well as in training, whilst those who only received it in training maintained a normal CPP. This suggests that insulin and leptin influence the retrieval of food reward associations rather than the initial formation of these associations. These results have been reinforced by studies which have shown decreased sucrose self-administration in response to insulin or leptin, and decreased sucrose licking following insulin.[16] The high circulating concentrations of insulin and leptin associated with obesity impair dopamine food reward pathways resulting in abnormal eating behaviours.
Summary of effects of centrally administered insulin and leptin on reward behaviours
| Behaviour | Effect of insulin | Effect of leptin |
|---|---|---|
| Brain self-stimulation | Decrease | Decrease |
| Relapse to heroin suckling | Not tested | Decrease |
| Acute sucrose suckling | Decrease | Not tested |
| Food-condtioned place preference | Decrease | Decrease |
| Sucrose self-administration | Decrease | Decrease |
| Acute chow intake | Decrease | Decrease |
| Opioid-stimulated sucrose | Decrease | Decrease |
- Adapted from Figlewicz et al. (2004)
Ghrelin can increase dopamine signalling on VTA neurones, via direct activation and also indirect manipulation of inputs onto the VTA to those of an excitatory nature. However, it is not clear whether this is a major part of the mechanism by which ghrelin stimulates feeding.[17]
Opioid and cannabinoid systems
Other reward systems, including the endogenous opioid and endocannabinoid systems interact with dopamine reward pathways. Opioid peptides act in the nucleus accumbens to increase ‘wanting’ and ‘liking’ of food rewards. [18]Opioids also influence mesolimibic dopamine pathways by inhibiting GABAergic input onto the dopamine neurones of the VTA, resulting in increased dopamine release [19]. The endocannabinoid system has also been implicated in reward behaviours, and cannabinoid receptors are expressed in several brain areas implicated in reward, including in the mesolimibic system. Endocannabinoids modulate neurotransmission, and, for example, cannabinoid agonists stimulate dopamine release. Conversly, antagonising these receptors inhibits activation of dopamine release. [20]
Food reward and obesity
There is a difference in dopamine activity between obese women and lean women/men in response to food and satiety. The obese have a higher metabolic activity in the parietal somatosensory area of the cortex which is linked to the sensory mouth, lips and tongue. Deficiencies of the dopamine D2 receptor may increase the likelihood of being obese. [21] Obese people have fewer D2 receptors in the striatum and with both the D1 and D2 receptors acting synergistically to decrease feeding, this altered expression causes increased eating. The DRD2 gene is responsible for the reinforcing properties of food/addictive behaviour. [22] Those who have the allelic variant A1 in this gene have fewer D2 receptors, making the dopamine reward circuits less sensitive.
References
- ↑ Berridge KC (2007) The debate over dopamine’s role in reward: the case for incentive salience. Psychopharmacology 191:391–431
- ↑ Changizi MA et al. (2002) Evidence that appetitive responses for dehydration and food-deprivation are learned Physiol Behav 75:295–304
- ↑ Wise RA (2006) Role of brain dopamine in food reward and reinforcement Phil Trans R Soc Lond B Biol Sci 361:1149–58
- ↑ Steiner JE et al.(2001) Comparative expression of hedonic impact: affective reactions to taste by human infants and other primates Neurosci Biobehav Rev 25:53–74
- ↑ Epstein LH et al.(2007) Food reinforcement and eating: a multilevel analysis Psychol Bull 133:884–906
- ↑ Liebman JM, Butcher LL (1974) Comparative involvement of dopamine and noradrenaline in rate-free self-stimulation in substantia nigra, lateral hypothalamus, and mesencephalic central gray N-S Arch Pharmacol
- ↑ Fouriezos G, Wise RA (1976) Pimozide-induced extinction of intracranial self-stimulation: response patterns rule out motor or performance deficits Brain Res 103:377–80
- ↑ Wickens JR et al. (2007) Striatal contributions to reward and decision making Ann NY Acad Sci 1104:192–212
- ↑ Delgado MR (2007) Reward-related responses in the human striatum Ann NY Acad Sci 1104: 70–88
- ↑ Natori S et al. (2009) Subsecond reward-related dopamine release in the mouse dorsal striatum Neurosci Res 63:267–72
- ↑ Darvas M, Palmiter RD (2009) Restriction of dopamine signaling to the dorsolateral striatum is sufficient for many cognitive behaviours PNAS 106;34:14664–9
- ↑ Palmiter RD(2008) Dopamine signaling in the dorsal striatum is essential for motivated behaviors: lessons from dopamine-deficient mice Ann N Y Acad Sci 1129:35–46
- ↑ Figlewicz DP, Benoit SC (2009) Insulin, leptin, and food reward: update 2008 Am J Physiol 296:R9-19
- ↑ Abizaid A et al. (2006) Ghrelin modulates the activity and synaptic input organization of midbrain dopamine neurones while promoting appetite J Clin Invest 116:3229–39
- ↑ Magni P et al. (2009) Feeding behavior in mammals including humans. Ann NY Acad Sci 1163:221-32
- ↑ Sipols AJ et al. (2000) Insulin and raclopride combine to decrease short-term intake of sucrose solutions. Peptides 21:1361–7
- ↑ Palmiter RD (2007) Is dopamine a physiologically relevant mediator of feeding behaviour? TINS 8:375-81
- ↑ Pecina S (2008) Opioid reward 'liking' and 'wanting' in the nucleus accumbens Physiol Behav 94:675-80
- ↑ Spanagel R, Weiss F (1999) The dopamine hypothesis of reward: past and current status TINS 22:521-7
- ↑ Solinas M (2008) The endocannabinoid system in brain reward processes Br J Pharmacol 154:369-83
- ↑ Wang et al. (2001) Brain dopamine and obesity. Lancet 357354-57
- ↑ Noble EP et al.(1994) D2 dopamine receptor gene and obesity. Int J Eating Disorders 15:205–17