Lipid
Template:Unreferenced Lipids are a class of hydrocarbon-containing organic compounds essential for the structure and function of living cells. Lipids are characterized by being water-insoluble and soluble in nonpolar organic solvents. Although the term lipid is often used as a synonym for fat, the latter is in fact a subgroup of lipids called triglycerides.
Types of lipids
Along with proteins, nucleic acids and carbohydrates, lipids are one of the major classes of biologically important molecules or biomolecules. However, unlike these other groups of molecules, lipids comprise a broader and more diverse range of structures. The term lipid really is a catch-all phrase for a wide variety of hydrocarbon-based molecules of biological origin that are predominantly nonpolar or hydrophobic ("water-fearing"), meaning that they do not interact well with polar solvents like water, and are instead soluble in less polar organic solvents. Most lipids also have some polar or hydrophilic ("water-loving") character. This makes them amphipathic or amphiphilic molecules (having both hydrophobic and hydrophilic portions). In the case of cholesterol, the polar group is a mere -OH (hydroxyl or alcohol). In the case of phospholipids, the polar groups are considerably larger and more polar. Lipids encompass a huge range of structures. They can be aliphatic or aromatic. They can be acyclic or cyclic, straight or branched, saturated or unsaturated. Lipids can be flexible or rigid. This diversity makes it impossible to define lipids on the basis of a single core structural feature or biosynthetic origin, as can be done with the other major groups of biomolecules. The basic classes of lipids are:
- Fatty acids
- Saturated
- Unsaturated
- Glycerides or glycerolipids
- Monoglycerides
- Diglycerides
- Triglycerides (neutral fats)
- Phosphoglycerides or glycerophospholipids
- Glycerol ethers
- Nonglycerides
- Sphingolipids
- Sterol lipids (includes cholesterol and steroid hormones)
- Prenol lipids (includes terpenoids)
- Waxes
- Polyketides
- More complex lipid derivatives
- Sugar-linked lipids: glycolipids
- Protein-linked lipids
Note: There are different ways to classify lipids. A comprehensive new classification system has been proposed recently (J. Lipid Res. 46:839), which instead divides lipids into: (1) fatty acyls, (2) glycerolipids, (3) glycerophospholipids, (4) sphingolipids, (5) sterol lipids, (6) prenol lipids, (7) saccharolipids and (8) polyketides.
Lipid functions
- Cell membrane structure
- Constitutes a barrier for the cell
- Controls membrane fluidity
- Controls the flow of material in and out of the cell
- Energy storage (for instance, fats stored in adipose tissue)
- Lipid hormones like steroids and eicosanoids - mediate communication between cells
- Signal transduction - function in the transmission of information in cells
- Lipid vitamins - required for metabolism, usually as coenzymes
Structure
Fatty acids and glycerides
Chemically, fatty acids can be described as long-chain monocarboxylic acids and have a general structure of CH3(CH2)nCOOH. The length of the chain usually ranges from 12 to 24, always with an even number of carbons. When the carbon chain contains no double bonds, it is called saturated. If it contains one or more such bonds, it is unsaturated. The presence of double bonds generally reduces the melting point of fatty acids. Furthermore, unsaturated fatty acids can occur either in cis or trans geometric isomers. In most naturally occurring fatty acids, the double bonds are in the cis configuration.
Glycerides are lipids possessing a glycerol core structure with one or more fatty acyl groups, which are fatty acid-derived chains attached to the glycerol backbone by ester linkages. Glycerides with three acyl groups (triglycerides or neutral fats) are the main storage form of fat in animals and plants.
An important type of glyceride-based molecule found in biological membranes, such as the cell's plasma membrane and the intracellular membranes of organelles, are the phosphoglycerides or glycerophospholipids. These are phospholipids that contain a glycerol core linked to two fatty acid-derived "tails" by ester or, more rarely, ether linkages and to one "head" group by a phosphate ester linkage. The head groups of the phospholipids found in biological membranes are phosphatidylcholine (lecithin), phosphatidylethanolamine, phosphatidylserine and phosphatidylinositol. These phospholipids are subject to a variety of reactions in the cell: for instance, polar head groups or fatty acid tails can be released from specific phospholipids through enzyme-catalyze hydrolysis to generate second messengers involved in signal transduction. In the case of phosphatidylinositol, the head group can be enzymatically modified by the addition of one to three phosphate molecules, and this constitutes another mechanism of cell signaling. While phospholipids are the major component of biological membranes, other non-glyceride lipid components like sphingolipids and sterols (such as cholesterol in animal cell membranes) are also found in biological membranes.
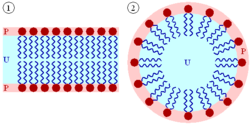
A biological membrane is a form of lipid bilayer, as is a liposome. Formation of lipid bilayers is a spontaneous process when the glycerophospholipids described above are placed in water. In an aqueous milieu, the polar heads of lipids tend to oriente toward the polar, aqueous environment, while the hydrophobic tails tend to minimize their contact with water. The nonpolar tails of lipids (U) tend to cluster together, forming a lipid bilayer (1) or a micelle (2). The polar heads (P) face the aqueous environment. Micelles form when single-tailed amphiphilic lipids are placed in a polar milieu, while lipid bilayers form when two-tailed phospholipids are placed in a polar environment (Fig. 2). Micelles are "monolayer" spheres and can only reach a certain size, whereas bilayers can be considerably larger. They can also form tubules. Bilayers that fold back upon themselves form a hollow sphere, enclosing a separate aqueous compartment, which is the basis of biological membranes.
Micelles and bilayers separate out from the polar milieu by a process known as the "hydrophobic effect." When dissolving a nonpolar substance in a polar environment, the polar molecules (i.e. water in an aqueous solution) become more ordered around the dissolved nonpolar substance, since the polar molecules cannot form hydrogen bonds to the nonpolar molecule. Therefore, in an aqueous environment, the polar water molecules form an ordered "clathrate" cage around the dissolved nonpolar molecule. However, when the nonpolar molecules separate out from the polar liquid, the entropy (state of disorder) of the polar molecules in the liquid increases. This is essentially a form of phase separation, similar to the spontaneous separation of oil and water into two separate phases when one puts them together.
The self-organisation depends on the concentration of the lipid present in solution. Below the critical micelle concentration, the lipids form a single layer on the liquid surface and are dispersed in solution. At the first critical micelle concentration (CMC-I), the lipids organise in spherical micelles, at the second critical micelle concentration (CMC-II) into elongated pipes, and at the lamellar point (LM or CMC-III) into stacked lamellae of pipes. The CMC depends on the chemical composition, mainly on the ratio of the head area and the tail length.
Nutrition and health
Lipids play diverse and important roles in nutrition and health. Many lipids are absolutely essential for life. However, there is also considerable awareness that abnormal levels of certain lipids, particularly cholesterol (in hypercholesterolemia) and, more recently, trans fatty acids, are risk factors for heart disease and other diseases.
References
- Chapter 12 in "Biochemistry" by Jeremy M. Berg, John L. Tymoczko and Lubert Stryer (2002) W. H. Freeman and Co.
- Alberts, B., et al. (2004) "Essential Cell Biology, 2nd Edition." Garland Science. ISBN 0-8153-3480-X
See also
External links
- ApolloLipids - Provides dyslipidemia and cardiovascular disease prevention and treatment information as well as continuing medical education programs.
- Lipids, Membranes and Vesicle Trafficking - The Virtual Library of Biochemistry and Cell Biology
- The Lipid library - provides information on the chemistry, analysis and biochemistry of lipids
- LIPID MAPS: LIPID Metabolites and Pathways Strategy
- IUPAC glossary entry for the lipid class of molecules what is IUPAC?
- "A comprehensive classification system for lipids"
- CCMDWeb, a lipid health educational resource on global risk reduction for dyslipidemia and cardiovascular disease as well as online CME programs.
ar:دسم bg:Липид ca:Lípid cs:Lipid da:Lipid de:Lipide es:Lípido eo:Lipido fa:لیپید fr:Lipide ko:지질 is:Fita it:Lipidi he:ליפיד lv:Lipīdi lb:Lipid lt:Riebalai mk:Липид nl:Lipide ja:脂質 pl:Lipid pt:Lípido ru:Липиды sk:Lipid sl:Lipid su:Lipid fi:Lipidi sv:Lipid th:ไลปิด vi:Lipit zh:脂類