Magnocellular neurosecretory cell
Magnocellular neurosecretory cells are neuroendocrine neurons whose cell bodies are mainly in the supraoptic nucleus and paraventricular nucleus of the hypothalamus. Some are also found in smaller numbers in accessory cell groups between these two nuclei, the largest one being the nucleus circularis. There are two types of magnocellular neurosecretory cells, oxytocin-producing cells and vasopressin-producing cells, but a few can produce both hormones. These cells are neuroendocrine neurons, they are electrically excitable, and generate action potentials in response to afferent stimulation.
Morphology
In the rat, where they have been most extensively studied, an estimated 10,000- 19,000 magnocellular neurosecretory cells project to the posterior pituitary gland, half of which contain vasopressin and half oxytocin. Each supraoptic nucleus contains about 2,000 magnocellular vasopressin neurons and about 1,000 magnocellular oxytocin neurons, and these are the only neurons present in the supraoptic nucleus. The paraventricular nucleus also contains many magnocellular neurons, but contains many other neuronal types in addition to these. It contains more (magnocellular) oxytocin neurons than vasopressin neurons,
However, nearly as many magnocellular neurons are dispersed as isolated cells or small groups throughout the anterior hypothalamus than are contained within the supraoptic and paraventricular nuclei together.
Each magnocellular neuron has a single long varicose axon which projects to the posterior pituitary with few if any collateral branches. Each axon when it reaches the posterior pituitary, gives rise to about 2,000 neurosecretory terminals and about 400 large axon swellings that store large numbers of hormone-containing vesicles. Each nerve terminal contains about 250 vesicles, and each swelling about 2,200 vesicles. The vesicles, each of which contains about 85,000 molecules of oxytocin or vasopressin, are released from the axon swellings and nerve terminals by exocytosis in response to calcium entry through voltage-gated channels, that occurs when action potentials are propagated down the axons.[2]
The cells have an interesting morphological association with glial cells - specialised astrocytes in the hypothalamus have long sheet-like processes that enclose "bundles" of dendrites, facilitating dendro-dendritic communication between cells.
Dendrites and afferent inputs
The cells typically have two or three long dendrites, which also contain large dilations, and which also contain a very high density of hormone-containing vesicles - each vasopressin-containing dendrite contains about 11,000 vesicles. Oxytocin and vasopressin can thus be released within the brain from these dendrites, as well as into the blood from the terminals in the posterior pituitary gland. However the release of oxytocin and vasopressin from dendrites is not consistently accompanied by peripheral secretion, as dendritic release is regulated differently. Dendritic release can be triggered by depolarisation, but can also be triggered by the mobilisation of intracellular calcium stores (in the endoplasmic reticulum).
The dendrites receive most of the synaptic inputs from afferent neurons that regulate the magnocellular neurons; typically a magnocellular neuron receives about 10,000 synapses from afferent neurons. Most of these inputs use either glutamate or GABA as a primary neurotransmitter, and many of these derive from anterior brain regions adjacent to the wall of the thrd ventricle - from the subfornical organ, the organum vasculosum of the lamina terminalis and from the nucleus medianus. However, there is also a prominent exctitatory noradrenaline noradrenergic innervation from the A1 cell group in the caudal ventrolateral medulla and from the A2 cell group in the nucleus of the solitary tract. Other inputs come from the arcuate nucleus and the olfactory bulb. Many of these afferent fibres contain neuropeptides as well as conventional transmitters, and, for example, somatostatin, angiotensin and several endogenous opioids are active on magnocellular neurons, which express specific receptors for a great many different peptides. Vasopressin cells and oxytocin cells differ in their sensitivity to many of these peptides; thus, for example, oxytocin cells are particular sensitive to mu-opioid agonists, to alpha-melanocyte stimulating hormone (via MC4 receptors) and to oxytocin itself; while vasopressin cells are sensitive to vasopressin. Both cell types are potently inhibited by kappa-opioid agonists. The cells also receive dopaminergic, cholinergic and histaminergic afferents.
Coexisting peptides
Magnocellular neurons make several other peptides in addition to oxytocin and vasopressin, though in much smaller amounts. The most abundant of these are cholecystokinin (made mainly in oxytocin neurons), galanin (made mainly in vasopressin cells) and the opioid peptide dynorphin (made in both cell types, but particularly in vasopressin cells). Other co-existing peptides include appelin and neuropeptide Y.
The neurons also express neuronal nitric oxide synthase, leading to activity-dependent production of nitric oxide which acts as a retrograde modulator of afferent inputs and a negative feedback mechanism to limit excessive electrical activity. They also produce endocannabinoids which also act as retrograde modulators of activity.
Intrinsic Properties
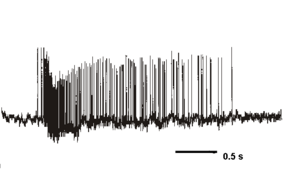
Oxytocin and vasopressin neurons have many intrinsic properties in common, but there are also some important differences. Both cell types fire relatively prolonged action potentials with a prominent calcium component mediated by high-voltage activated calcium conductances. This large calcium entry is buffered differently in the two cell types, which express different calcium-binding proteins. However, in both cells, spike activity leads to the activation of several types of voltage- and calcium sensitive potassium channels, leading to (i) a post-spike hyperpolarising afterpotential (HAP), lasting typically about 50 ms, and (ii) a slower afterhyperpolarisation (AHP), lasting for several sseconds which follows bursts of spike activity. In addition, vasopressin neurons express an activity-dependent slow depolarisation, called a depolarising afterpotential (DAP); the HAP, AHP and DAP overlap in time, so that spikes in vasopressin cells are followed by a sequence of hyperpolarisation, depolarisation and late hyperpolarisation. As the DAPs summate with repeated spike activity they sustain prolonged activity-dependent plateau potentials that underlie repeated long bursts of action potentials.
Osmosensitivity
Magnocellular neurons sense and respond to plasma osmolality by both intrinsic and extrinsic mechanisms. When the osmotic pressure of the extracellular fluid increases (or decreases), all cells that have membrane water channels will shrink (or swell) as water leaves (or enters). Most cells correct this volume change via cell volume regulatory mechanisms, including by exporting neutral osmolytes such as the amino acid taurine. However, magnocellular neurons do not rapidly correct their volume changes, and when they shrink in response to an acute hyperosmotic challenge, the decrease in membrane stretch leads to a sustained depolarization via opening of non-inactivating, stretch-inactivated non-selective cation channels. Conversely, when they swell, as in response to a hypo-osmotic challenge, this leads to sustained hyperpolarisation via the closure of these channels.
Magnocellular neurons are thus directly osmosensitive (in the rat at least), but this mechanism is not sufficient to trigger increased vasopressin and oxytocin secretion. These neurons receive inputs from several brain areas that relay information about the hydration state of the animal. The most important of these areas is the region ventral to the anterior wall of the third ventricle, lesions to which can destroy the ability of magnocellular neurons to respond to increased osmolality. This region contains the nucleus medianus, and two circumventricular organs, the organum vasculosum of the lamina terminalis and the subfornical organ, both of which project directly to the magnocellular neurons and transmits information about plasma osmolality, and also about circulating factors including the peptides angiotensin and relaxin. These projections include both excitatory and inhibitory components; and an increase in systemic osmolality causes increased release of both GABA and glutamate within the supraoptic nucleus.
References
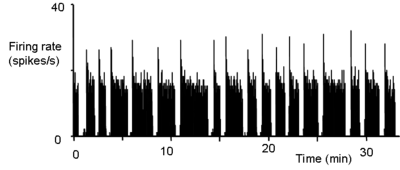
- ↑ Dyball REJ, Leng G (1986) Regulation of the milk ejection reflex in the rat. J Physiol 380:239-56. PMID 3612564
- ↑ The detailed ultrastructural measurements were made independently by Jean Nordmann in Strasbourg and John Morris in Bristol. Reviewed in Leng G, Ludwig M (2008) Neurotransmitters and peptides: whispered secrets and public announcements.J Physiol 586:5625-32 PMID 18845614