User:Milton Beychok/Sandbox
The heat of vaporization, ( or ) is the amount of thermal energy required to convert a quantity of liquid into a vapor. It can be thought of as the energy required to break the intermolecular bonds within the liquid.
It is also often referred to as the latent heat of vaporization ( or ) and the enthalpy of vaporization ( or or ) and is usually measured and reported at the temperature corresponding to the normal boiling point of the liquid. Sometimes reported values have been corrected to a temperature of 298 K.
Measurement units
Heat of vaporization values are usually reported in measurement units such as J/mol or kJ/mol and referred to as the molar heat of vaporization, although J/g or kJ/kg are also often used. Older units such as kcal/mol, cal/g, Btu/lb and others are still used sometimes.
Temperature dependency
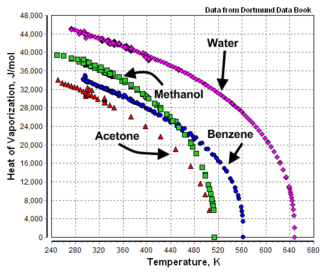
The heat of vaporization is not a constant. It is temperature dependent as shown in Figure 1 by the example graphs of temperature versus heat of vaporization for acetone, benzene, methanol and water.
As shown by the example graphs, the heat of vaporization of a liquid at a given temperature (other than the normal boiling point temperature) may vary significantly from the value reported at the normal boiling point of the liquid.
Estimating heat of vaporization values
| Name | Formula | Hv | Tn | Tc | Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| ( J/mol ) | ( °C ) | ( K ) | ( K ) | ( atm ) | |||||
| Acetic acid | C2H4O2 | 23,700 | 117.9 | 391.1 | 594.8 | 57.1 | |||
| Acetone | C3H6O | 29,100 | 56.2 | 329.4 | 508.7 | 47.0 | |||
| Benzene | C6H6 | 30,720 | 80.0 | 353.2 | 562.1 | 48.6 | |||
| Butane | C4H10 | 22,440 | – 0.5 | 272.7 | 425.2 | 37.5 | |||
| Carbon tetrachloride | CCl4 | 29,820 | 76.6 | 349.8 | 556.3 | 45.0 | |||
| Chloroform | CHCl3 | 29,240 | 61.1 | 334.3 | 536.2 | 54.0 | |||
| Cyclohexane | C6H12 | 29,970 | 80.7 | 353.9 | 553.6 | 40.0 | |||
| Cyclopentane | C5H10 | 27,300 | 49.2 | 322.4 | 511.8 | 44.6 | |||
| Decane | C10H22 | 38,750 | 174.1 | 447.3 | 617.6 | 20.8 | |||
| Ethanol | C2H6O | 38,560 | 78.2 | 351.4 | 516.2 | 63.0 | |||
| Hexane | C6H14 | 28.850 | 68.7 | 341.9 | 507.4 | 29.9 | |||
| Isobutane | C4H10 | 21,300 | – 11.9 | 261.3 | 408.2 | 36.0 | |||
| Methanol | CH4O | 35,210 | 64.7 | 337.9 | 513.2 | 78.5 | |||
| Octane | C8H18 | 34,410 | 125.6 | 398.8 | 569.2 | 24.8 | |||
| Water | H20 | 40,660 | 100 | 373.2 | 647.3 | 218.3 | |||
Notes:
| |||||||||
References
- ↑ Dortmund Data Bank Online Search
- ↑ J.M. Smith, H.C. Van Ness and N.M. Abbot (2004). Introduction to Chemical Engineering Thermodynamics, 7th Edition. McGraw-Hill. ISBN 0-07-310445-0.
- ↑ Robert C. Weast (Editor) (1976). Perry's Chemical Engineers' Handbook, 56th Edition. CRC Press. ISBN 0-87819-455-X.