User:Milton Beychok/Sandbox: Difference between revisions
imported>Milton Beychok No edit summary |
imported>Milton Beychok No edit summary |
||
| Line 1: | Line 1: | ||
{{Image|Hydrometer.png|right|223px|A schematic of a specific gravity hydrometer}} | {{Image|Hydrometer.png|right|223px|A schematic of a specific gravity hydrometer}} | ||
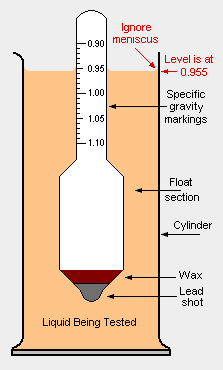
A '''hydrometer''' is an instrument used to measure the [[specific gravity]] (or ''relative density'') of [[Liquid/Definition|liquid]]s; that is, the ratio of the [[Density (chemistry)|density]] of the liquid to the density of [[water]] with both at the same [[temperature]]. It is usually made of glass and consists of a small diameter cylindrical stem and a larger diameter section weighted with [[mercury]] or [[Lead|lead shot]] (sealed with [[wax]]) to make it float upright. The larger diameter section is necessary to provide the displacement volume needed for the proper [[buoyancy]] of the hydrometer | A '''hydrometer''' is an instrument used to measure the [[specific gravity]] (SG) (or ''relative density'') of [[Liquid/Definition|liquid]]s; that is, the ratio of the [[Density (chemistry)|density]] of the liquid to the density of [[water]] with both at the same [[temperature]]. It is usually made of glass and consists of a small diameter cylindrical stem and a larger diameter section weighted with [[mercury]] or [[Lead|lead shot]] (sealed with [[wax]]) to make it float upright. The larger diameter section is necessary to provide the displacement volume needed for the proper [[buoyancy]] of the hydrometer | ||
The stem contains a rolled paper marked with the scale being used. There are a great many different hydrometer scales commonly used to measure liquid densities in: [[petroleum crude oil]] marketing and refining; making [[wine]], brewing [[beer]] and making [[whiskey]]; producing [[sulfuric acid]] and other industrial [[chemical]]s; and refining [[sugar]]. | The stem contains a rolled paper marked with the scale being used. There are a great many different hydrometer scales commonly used to measure liquid densities in: [[petroleum crude oil]] marketing and refining; making [[wine]], brewing [[beer]] and making [[whiskey]]; producing [[sulfuric acid]] and other industrial [[chemical]]s; and refining [[sugar]]. | ||
| Line 9: | Line 9: | ||
The many hydrometer scales that are commonly used include: | The many hydrometer scales that are commonly used include: | ||
; | ;Specific gravity (SG): This scale is widely used in [[chemistry]], [[engineering]] and [[physics]] as well as by [[geologist]]s, [[mineralogist]]s and [[gemologist]]s. It is often referred to as ''relative density'' and it is the one shown in the adjacent drawing. Specific gravity is expressed as: | ||
:<math>\mathrm{SG}_{4\,^\circ \mathrm{C}}^{4\,^\circ \mathrm{C}} = \frac {\mathrm{density\; of\; the\; tested\; liquid\; at\; 4\,^\circ \mathrm{C}}} {\mathrm{density\; of\; water\; at\; 4\,^\circ \mathrm{C}}}</math> | :<math>\mathrm{SG}_{4\,^\circ \mathrm{C}}^{4\,^\circ \mathrm{C}} = \frac {\mathrm{density\; of\; the\; tested\; liquid\; at\; 4\,^\circ \mathrm{C}}} {\mathrm{density\; of\; water\; at\; 4\,^\circ \mathrm{C}}}</math> | ||
| Line 15: | Line 15: | ||
:Unless the two reference temperatures are explicitly stated, they are generally taken to be 4 [[Celsius (unit)|°C]]. | :Unless the two reference temperatures are explicitly stated, they are generally taken to be 4 [[Celsius (unit)|°C]]. | ||
;[[API gravity]]: This scale was developed by the [[American Petroleum Institute]] (API) in 1921 for use in the petroleum industry. It is expressed as: | ;[[API gravity]]: This scale was developed by the [[American Petroleum Institute]] (API) in 1921 for use in the [[petroleum industry]] and it is now universally used by the petroleum industry worldwide. It is expressed as: | ||
:<math>^\circ \mathrm{API} = \frac{141.5}{\mathrm{SG}_{60\,^\circ \mathrm{F}}^{60\,^\circ \mathrm{F}}} - \mathrm{131.5}</math> and <math>\mathrm{SG}_{60\,^\circ \mathrm{F}}^{60\,^\circ \mathrm{F}} = \frac {\mathrm{density\; of\; petroleum\; liquid\; at\; 60\,^\circ \mathrm{F}}} {\mathrm{density\; of\; water\; at\; 60\,^\circ \mathrm{F}}}</math> | :<math>^\circ \mathrm{API} = \frac{141.5}{\mathrm{SG}_{60\,^\circ \mathrm{F}}^{60\,^\circ \mathrm{F}}} - \mathrm{131.5}</math> and <math>\mathrm{SG}_{60\,^\circ \mathrm{F}}^{60\,^\circ \mathrm{F}} = \frac {\mathrm{density\; of\; petroleum\; liquid\; at\; 60\,^\circ \mathrm{F}}} {\mathrm{density\; of\; water\; at\; 60\,^\circ \mathrm{F}}}</math> | ||
| Line 21: | Line 21: | ||
:Note: 60 [[Fahrenheit (unit)|°F]] is equivalent to 15.56 °C. | :Note: 60 [[Fahrenheit (unit)|°F]] is equivalent to 15.56 °C. | ||
;[[Baumé gravity]]: These two scales, one for liquids lighter than water and one for liquids heavier than water, | ;[[Baumé gravity]]: These two scales, one for liquids lighter than water and one for liquids heavier than water, were developed by the [[France|French]] chemist [[Antoine Baumé]] in 1768. It is widely used in industrial chemistry, [[pharmacology, [[sugar refining]] and other industries. The two scales are are expressed as: | ||
:<math>^\circ \mathrm{B\acute{e}} = \frac{140}{\mathrm{SG}_{20\,^\circ \mathrm{C}}^{20\,^\circ \mathrm{C}}} - \mathrm{130}</math> (for liquids lighter than water) and <math>\mathrm{SG}_{20\,^\circ \mathrm{C}}^{20\,^\circ \mathrm{C}} = \frac {\mathrm{density\; of\; the\; tested\; liquid\; at\; 20\,^\circ \mathrm{C}}} {\mathrm{density\; of\; water\; at\; 20\,^\circ \mathrm{C}}}</math> | :<math>^\circ \mathrm{B\acute{e}} = \frac{140}{\mathrm{SG}_{20\,^\circ \mathrm{C}}^{20\,^\circ \mathrm{C}}} - \mathrm{130}</math> (for liquids lighter than water) and <math>\mathrm{SG}_{20\,^\circ \mathrm{C}}^{20\,^\circ \mathrm{C}} = \frac {\mathrm{density\; of\; the\; tested\; liquid\; at\; 20\,^\circ \mathrm{C}}} {\mathrm{density\; of\; water\; at\; 20\,^\circ \mathrm{C}}}</math> | ||
Revision as of 21:49, 2 February 2010
A hydrometer is an instrument used to measure the specific gravity (SG) (or relative density) of liquids; that is, the ratio of the density of the liquid to the density of water with both at the same temperature. It is usually made of glass and consists of a small diameter cylindrical stem and a larger diameter section weighted with mercury or lead shot (sealed with wax) to make it float upright. The larger diameter section is necessary to provide the displacement volume needed for the proper buoyancy of the hydrometer
The stem contains a rolled paper marked with the scale being used. There are a great many different hydrometer scales commonly used to measure liquid densities in: petroleum crude oil marketing and refining; making wine, brewing beer and making whiskey; producing sulfuric acid and other industrial chemicals; and refining sugar.
Scales
The many hydrometer scales that are commonly used include:
- Specific gravity (SG)
- This scale is widely used in chemistry, engineering and physics as well as by geologists, mineralogists and gemologists. It is often referred to as relative density and it is the one shown in the adjacent drawing. Specific gravity is expressed as:
- Unless the two reference temperatures are explicitly stated, they are generally taken to be 4 °C.
- API gravity
- This scale was developed by the American Petroleum Institute (API) in 1921 for use in the petroleum industry and it is now universally used by the petroleum industry worldwide. It is expressed as:
- and
- Note: 60 °F is equivalent to 15.56 °C.
- Baumé gravity
- These two scales, one for liquids lighter than water and one for liquids heavier than water, were developed by the French chemist Antoine Baumé in 1768. It is widely used in industrial chemistry, [[pharmacology, sugar refining and other industries. The two scales are are expressed as:
- (for liquids lighter than water) and
- (for liquids heavier than water) and
History
Knowledge of relative density or specific gravity has been with us since the days of Archimedes in 250 BC, with the observation that light objects can float while heavier ones will sink in water.[1] Availa Hypatia (born ca. 350 and died 415 AD), a Greek scholar from Alexandria in Egypt, is reputed to have invented the hydrometer.[2]
References
- ↑ T. L. Heath (1897). The Works of Archimedes. Cambridge University Press, page 253. Full text available at www.archive.org
- ↑ Mothers of Invention Ethlie Ann Vare and Greg Ptacek, 1988, pp. 24-26.