Chemical bond: Difference between revisions
imported>Paul Wormer |
imported>Paul Wormer No edit summary |
||
| Line 8: | Line 8: | ||
* Intermolecular (also known as Van der Waals) bonds. These are bonds between stable molecules, see the articles [[intermolecular forces]] and [[Van der Waals forces]] for more details. For many years it was believed that [[hydrogen bond]]ing should be classified as a separate type of bond, but modern theoretical chemistry recognizes it as a special type of intermolecular bond. | * Intermolecular (also known as Van der Waals) bonds. These are bonds between stable molecules, see the articles [[intermolecular forces]] and [[Van der Waals forces]] for more details. For many years it was believed that [[hydrogen bond]]ing should be classified as a separate type of bond, but modern theoretical chemistry recognizes it as a special type of intermolecular bond. | ||
It took several centuries before chemical bonding was fully understood, but at present it is generally accepted that non-relativistic quantum mechanical explanations based on Coulomb's electrostatic law<ref>That is, explanations derived from quantum mechanical energy operators containing electron-electron, electron-nucleus, and nucleus-nucleus Coulomb interactions plus electronic kinetic energies.</ref> give satisfactory accounts of all kinds of bonds.<ref>As late as 1916 the famous American chemist [[Gilbert N. Lewis]] disagreed strongly with this statement. He saw electrons as stationary without kinetic energy and not exerting Coulomb forces. In a lecture given at the December meeting of the Sections of Physics and Chemistry of the [[American Association for the Advancement of Science]], the [[American Physical Society]], and the [[American Chemical Society]] (see Science Magazine pp. 297-302 (1917); [http://dx.doi.org/10.1126/science.46.1187.297 DOI]), he declared the following: "Therefore, unless we are willing, under the onslaught of quantum theories, to throw overboard all of the basic principles of physical science, we must conclude that the electron in the Bohr atom not only ceases to obey Coulomb's law, but exerts no influence whatsoever upon another charged particle at any distance." </ref> | It took several centuries before chemical bonding was fully understood, but at present it is generally accepted that non-relativistic quantum mechanical explanations based on Coulomb's electrostatic law<ref>That is, explanations derived from quantum mechanical energy operators containing electron-electron, electron-nucleus, and nucleus-nucleus Coulomb interactions plus electronic kinetic energies.</ref> give satisfactory accounts of all kinds of bonds.<ref>As late as 1916 the famous American chemist [[Gilbert Newton Lewis|G. N. Lewis]] disagreed strongly with this statement. He saw electrons as stationary without kinetic energy and not exerting Coulomb forces. In a lecture given at the December meeting of the Sections of Physics and Chemistry of the [[American Association for the Advancement of Science]], the [[American Physical Society]], and the [[American Chemical Society]] (see Science Magazine pp. 297-302 (1917); [http://dx.doi.org/10.1126/science.46.1187.297 DOI]), he declared the following: "Therefore, unless we are willing, under the onslaught of quantum theories, to throw overboard all of the basic principles of physical science, we must conclude that the electron in the Bohr atom not only ceases to obey Coulomb's law, but exerts no influence whatsoever upon another charged particle at any distance." </ref> | ||
Gravitational forces, strong nuclear forces, or even magnetic forces, do not play any significant role in chemical bonding. | Gravitational forces, strong nuclear forces, or even magnetic forces, do not play any significant role in chemical bonding. | ||
==Octet rule== | ==Octet rule== | ||
| Line 22: | Line 22: | ||
{{Image|Octet rule.png|right|350px|Examples of octet rule.}} | {{Image|Octet rule.png|right|350px|Examples of octet rule.}} | ||
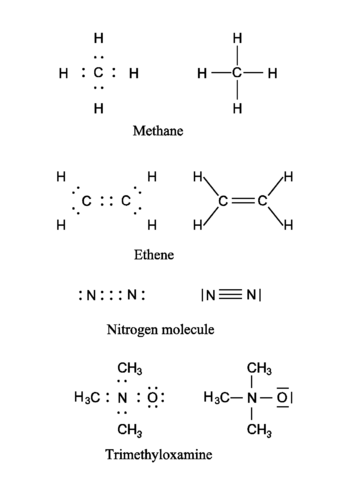
In the figure four molecules are shown as examples. Both the Lewis representation (electrons as dots) and the corresponding traditional chemical representation (chemical bonds as lines) are shown. In the first molecule (methane, CH<sub>4</sub>) the hydrogen atoms have a helium electronic configuration (are surrounded by two electrons) and the carbon has a neon configuration (is surrounded by an octet consisting of eight electrons). In the computation of the formal charge on the atoms it is assumed that the electrons in a bond are shared equally by the bonding partners. Thus, in methane the hydrogen atoms have one electron, while the carbon atom has four. From the above table it follows that the hydrogens and the carbon are formally neutral in CH<sub>4</sub>. The ethene (formerly known as ethylene) molecule shows a double bond between the carbon atoms. Note that both carbons have a neon configuration (are surrounded by four electron pairs). The next molecule (the nitrogen molecule N<sub>2</sub>) shows a triple bond. Both N-atoms are surrounded by an octet (four electron pairs). Each N-atom has five electrons and is formally neutral. The last molecule (trimethyloxamine) has as central atom a nitrogen atom with four electrons, and hence the nitrogen lacks an electron and has a formal charge +1. The oxygen atom has three pairs to itself and shares one pair, carrying seven electrons, so that oxygen has the formal charge −1. The total trimethyloxamine molecule is neutral. | In the figure four molecules are shown as examples. Both the Lewis representation (electrons as dots) and the corresponding traditional chemical representation (chemical bonds as lines) are shown. In the first molecule (methane, CH<sub>4</sub>) the hydrogen atoms have a helium electronic configuration (are surrounded by two electrons) and the carbon has a neon configuration (is surrounded by an octet consisting of eight electrons). In the computation of the formal charge on the atoms it is assumed that the electrons in a bond are shared equally by the bonding partners. Thus, in methane the hydrogen atoms have one electron, while the carbon atom has four. From the above table it follows that the hydrogens and the carbon are formally electrically neutral in CH<sub>4</sub>. The ethene (formerly known as ethylene) molecule shows a double bond between the carbon atoms. Note that both carbons have a neon configuration (are surrounded by four electron pairs). The next molecule (the nitrogen molecule N<sub>2</sub>) shows a triple bond. Both N-atoms are surrounded by an octet (four electron pairs). Each N-atom has five electrons and is formally neutral. The last molecule (trimethyloxamine) has as central atom a nitrogen atom with four electrons, and hence the nitrogen lacks an electron and has a formal charge +1. The oxygen atom has three pairs to itself and shares one pair, carrying seven electrons, so that oxygen has the formal charge −1. The total trimethyloxamine molecule is neutral. | ||
===Historical note=== | |||
The octet rule is usually attributed to [[Gilbert Newton Lewis]],<ref>G. N. Lewis, ''The Atom and the Molecule'', Journal American Chemical Society, vol. '''38''', pp. 762–785 (1916)</ref> who referred to it as the "rule of eight". The term "octet rule" is due to [[Irving Langmuir]], <ref>I. Langmuir, ''The Arrangement of Electrons in Atoms and Molecules'', Journal American Chemical Society, vol. '''41''', pp. 868–934 (1919)</ref> who extended and refined the work of Lewis. Lewis saw an atom as a cube with—depending on the atom—one to eight valence electrons on its corners. In this picture molecules consist of cubes piled on top of each other, or next to each other sharing a side or a corner. In Ref.<ref>W. B. Jensen, ''Abegg, Lewis, Langmuir, and the octet rule'', Journal Chemical Education, vol. '''61''', pp. 191–200 (1984); [http://dx.doi.org/10.1021/ed061p191 DOI]</ref> it is argued that this idea is not as novel as it is usually believed, but that the idea of eight valences was floating among contemporary chemists and that, for instance, the German chemist [[Richard Abegg]] presented already in 1902 some bonding concepts that came close to Lewis's 1916 ideas. In any case, it is generally accepted that Lewis was the first to see the importance of electron pairing. Lewis made the simple assumption that all atomic "kernels" (nuclei plus inner shell electrons) repel one another, and that molecules are held together only by the pairs of electrons which are held jointly by the component atoms. | |||
It is remarkable how far physics and chemistry still were separated during the first two decades of the twentieth century. Neither Lewis nor Langmuir put much faith in the [[Ernest Rutherford|Rutherford]]-[[Bohr]] planetary model of the atom in which the negative electrons orbit a positive nucleus. Both chemists believed electrons to be stationary on the vertices of cubic atoms. On the other hand, the founding fathers of quantum mechanical bonding theory (the physicists [[Walter Heitler]], [[Fritz London]], [[Georg Rumer]], [[Edward Teller]], and others) completely ignored the chemists (or perhaps were not even aware of their work). [[Linus Pauling]] deserves the credit for having bridged the two branches of science, which he did in a series of papers in the early 1930s and in his famous 1939 book ''The Nature of the Chemical Bond''<ref>L. Pauling, ''The Nature of the Chemical Bond'', Cornell University Press, 3rd edition, 1960</ref> that is dedicated to G. N. Lewis. In modern chemistry textbooks, chemical bonding theory is usually presented as a continuous development from Lewis's concepts to the Heitler-London theory for the binding in H<sub>2</sub>. Historically, such a presentation of the developments is an oversimplification. | |||
'''(To be continued)''' | '''(To be continued)''' | ||
== | ==Notes== | ||
<references /> | <references /> | ||
Revision as of 04:55, 20 April 2010
In chemistry, a chemical bond is a force between two atoms that is strong enough to see the two atoms that are exerting the force on each other as an entity. It can happen that the two atoms form a stable entity (a diatomic molecule), an example being two nitrogen atoms chemically bound to the stable molecule N—N (written as N2). It can also happen that the two atoms are part of a larger aggregate. For instance, the chemical bond C—O between a carbon (C) atom and an oxygen (O) atom in the molecule methanol H3C—OH is a strong and easily recognizable bond. Two atoms may also be bound in a crystal, such as sodium (Na) and chlorine (Cl) that appear in a 1-1 ratio in crystalline rock (kitchen) salt (NaCl). A diamond crystal can be seen as one huge molecule consisting of bonded C—C (carbon-carbon) pairs.
Traditionally one distinguishes in chemistry the following types of bonds:
- Covalent bonds. A covalent bond is an electron pair shared by two bonded atoms. These are the bonds most commonly found in organic chemistry. They take place mainly between hydrogen, carbon, nitrogen, and oxygen and lead to stable, recognizable, molecules that remain intact in the solid, liquid, and gaseous aggregation states. The phenomenon of electron pairing (covalent bonding) requires quantum mechanics for an explanation and deeper understanding.
- Ionic bonds. Here atom A loses an electron to its bonding partner B, so that A becomes the cation A+ and B the anion B−. Consecutively, the ions bind strongly through the Coulomb interaction. Systems of which the atoms are bound by ionic interactions are usually crystals, the example of rock salt (Na+—Cl−) was already mentioned. It requires advanced laboratory techniques to separate ionically bound molecules from crystals, because the crystals are very stable.
- Metallic bonds. A number of metal atoms can crystallize to form a metal, which is a solid recognized by high electric and thermal conductivity. The bonding is caused by delocalized electrons forming electronic bands. The mechanism is akin to the formation of molecular orbitals in molecules. An explanation of metallic bonding is offered by quantum mechanics. Metal molecules (M2, M3, etc.) are not easily prepared experimentally because the solids are usually very stable.
- Intermolecular (also known as Van der Waals) bonds. These are bonds between stable molecules, see the articles intermolecular forces and Van der Waals forces for more details. For many years it was believed that hydrogen bonding should be classified as a separate type of bond, but modern theoretical chemistry recognizes it as a special type of intermolecular bond.
It took several centuries before chemical bonding was fully understood, but at present it is generally accepted that non-relativistic quantum mechanical explanations based on Coulomb's electrostatic law[1] give satisfactory accounts of all kinds of bonds.[2] Gravitational forces, strong nuclear forces, or even magnetic forces, do not play any significant role in chemical bonding.
Octet rule
The octet rule is a simple rule that describes the valency of light atoms (atomic number Z ≤ 18, first and second row of the periodic system). The valency of an atom is the number of covalent bonds that the atom can make with a partner. The octet rule starts with the assumption that valency involves only the electrons in the outer shells of the (lowest-energy-state) atoms participating in the bonding. The following lists the number of valence electrons of the first 18 chemical elements
| H(1) | He(2) | ||||||
| Li(1) | Be(2) | B(3) | C(4) | N(5) | O(6) | F(7) | Ne(8) |
| Na(1) | Mg(2) | Al(3) | Si(4) | P(5) | S(6) | Cl(7) | Ar(8) |
The assumption is that the electron configuration of a noble gas (He, Ne, Ar) is particularly stable (since these gases are chemically inert) and that a bonded atom strives toward a noble gas configuration. Except for hydrogen, which strives for for a helium configuration of two electrons (a duplet), it means that an atom tries to surround himself with an octet of eight electrons. Of course electrons must be shared with bonding partners to achieve a noble gas configuration.
In the figure four molecules are shown as examples. Both the Lewis representation (electrons as dots) and the corresponding traditional chemical representation (chemical bonds as lines) are shown. In the first molecule (methane, CH4) the hydrogen atoms have a helium electronic configuration (are surrounded by two electrons) and the carbon has a neon configuration (is surrounded by an octet consisting of eight electrons). In the computation of the formal charge on the atoms it is assumed that the electrons in a bond are shared equally by the bonding partners. Thus, in methane the hydrogen atoms have one electron, while the carbon atom has four. From the above table it follows that the hydrogens and the carbon are formally electrically neutral in CH4. The ethene (formerly known as ethylene) molecule shows a double bond between the carbon atoms. Note that both carbons have a neon configuration (are surrounded by four electron pairs). The next molecule (the nitrogen molecule N2) shows a triple bond. Both N-atoms are surrounded by an octet (four electron pairs). Each N-atom has five electrons and is formally neutral. The last molecule (trimethyloxamine) has as central atom a nitrogen atom with four electrons, and hence the nitrogen lacks an electron and has a formal charge +1. The oxygen atom has three pairs to itself and shares one pair, carrying seven electrons, so that oxygen has the formal charge −1. The total trimethyloxamine molecule is neutral.
Historical note
The octet rule is usually attributed to Gilbert Newton Lewis,[3] who referred to it as the "rule of eight". The term "octet rule" is due to Irving Langmuir, [4] who extended and refined the work of Lewis. Lewis saw an atom as a cube with—depending on the atom—one to eight valence electrons on its corners. In this picture molecules consist of cubes piled on top of each other, or next to each other sharing a side or a corner. In Ref.[5] it is argued that this idea is not as novel as it is usually believed, but that the idea of eight valences was floating among contemporary chemists and that, for instance, the German chemist Richard Abegg presented already in 1902 some bonding concepts that came close to Lewis's 1916 ideas. In any case, it is generally accepted that Lewis was the first to see the importance of electron pairing. Lewis made the simple assumption that all atomic "kernels" (nuclei plus inner shell electrons) repel one another, and that molecules are held together only by the pairs of electrons which are held jointly by the component atoms.
It is remarkable how far physics and chemistry still were separated during the first two decades of the twentieth century. Neither Lewis nor Langmuir put much faith in the Rutherford-Bohr planetary model of the atom in which the negative electrons orbit a positive nucleus. Both chemists believed electrons to be stationary on the vertices of cubic atoms. On the other hand, the founding fathers of quantum mechanical bonding theory (the physicists Walter Heitler, Fritz London, Georg Rumer, Edward Teller, and others) completely ignored the chemists (or perhaps were not even aware of their work). Linus Pauling deserves the credit for having bridged the two branches of science, which he did in a series of papers in the early 1930s and in his famous 1939 book The Nature of the Chemical Bond[6] that is dedicated to G. N. Lewis. In modern chemistry textbooks, chemical bonding theory is usually presented as a continuous development from Lewis's concepts to the Heitler-London theory for the binding in H2. Historically, such a presentation of the developments is an oversimplification.
(To be continued)
Notes
- ↑ That is, explanations derived from quantum mechanical energy operators containing electron-electron, electron-nucleus, and nucleus-nucleus Coulomb interactions plus electronic kinetic energies.
- ↑ As late as 1916 the famous American chemist G. N. Lewis disagreed strongly with this statement. He saw electrons as stationary without kinetic energy and not exerting Coulomb forces. In a lecture given at the December meeting of the Sections of Physics and Chemistry of the American Association for the Advancement of Science, the American Physical Society, and the American Chemical Society (see Science Magazine pp. 297-302 (1917); DOI), he declared the following: "Therefore, unless we are willing, under the onslaught of quantum theories, to throw overboard all of the basic principles of physical science, we must conclude that the electron in the Bohr atom not only ceases to obey Coulomb's law, but exerts no influence whatsoever upon another charged particle at any distance."
- ↑ G. N. Lewis, The Atom and the Molecule, Journal American Chemical Society, vol. 38, pp. 762–785 (1916)
- ↑ I. Langmuir, The Arrangement of Electrons in Atoms and Molecules, Journal American Chemical Society, vol. 41, pp. 868–934 (1919)
- ↑ W. B. Jensen, Abegg, Lewis, Langmuir, and the octet rule, Journal Chemical Education, vol. 61, pp. 191–200 (1984); DOI
- ↑ L. Pauling, The Nature of the Chemical Bond, Cornell University Press, 3rd edition, 1960