Hepatocellular cancer: Difference between revisions
imported>Robert Badgett m (→Screening) |
imported>Anthony.Sebastian (testing) |
||
| Line 2: | Line 2: | ||
==Diagnosis== | ==Diagnosis== | ||
The [[alpha-fetoprotein]] may be elevated; however, its [[positive predictive value]] is low and it may be elevated in patients with [[cirrhosis]].<ref name="pmid11350553" | The [[alpha-fetoprotein]] may be elevated; however, its [[positive predictive value]] is low and it may be elevated in patients with [[cirrhosis]].<ref name="pmid11350553"/> | ||
==Treatment== | ==Treatment== | ||
{{PDQ-treatment|http://www.cancer.gov/cancertopics/pdq/treatment/adult-primary-liver/HealthProfessional/page5}} | {{PDQ-treatment|http://www.cancer.gov/cancertopics/pdq/treatment/adult-primary-liver/HealthProfessional/page5}} | ||
| Line 15: | Line 13: | ||
==Screening== | ==Screening== | ||
"Surveillance is deemed cost-effective if the expected HCC risk exceeds 1.5% per year in patients with hepatitis C and 0.2% per year in patients with hepatitis B. Analysis of recent studies show that alpha-fetoprotein determination lacks adequate sensitivity and specificity for effective surveillance (and for diagnosis). Thus, surveillance has to be based on ultrasound examination. The recommended screening interval is 6 months" according to a [[clinical practice guideline]] by the [http://www.aasld.org/ American Association for the Study of Liver Diseases]. <ref name="pmid21374666">{{cite journal| author=Bruix J, Sherman M, American Association for the Study of Liver Diseases| title=Management of hepatocellular carcinoma: an update. | journal=Hepatology | year= 2011 | volume= 53 | issue= 3 | pages= 1020-2 | pmid=21374666 | doi=10.1002/hep.24199 | pmc=PMC3084991 | url= }} </ref> | "Surveillance is deemed cost-effective if the expected HCC risk exceeds 1.5% per year in patients with hepatitis C and 0.2% per year in patients with hepatitis B. Analysis of recent studies show that alpha-fetoprotein determination lacks adequate sensitivity and specificity for effective surveillance (and for diagnosis). Thus, surveillance has to be based on ultrasound examination. The recommended screening interval is 6 months" according to a [[clinical practice guideline]] by the [http://www.aasld.org/ American Association for the Study of Liver Diseases]. <ref name="pmid21374666"/> | ||
"In a mixed-aetiology cohort, the most effective surveillance strategy is to screen each patient with AFP assay and ultrasound imaging on a 6-monthly basis" according to a [[systematic review]] by the [http://www.hta.ac.uk/ NIHR Health Technology Assessment programme] (UK). <ref name="pmid17767898"/> | |||
In a [[randomized controlled trial]], the [[relative risk ratio]] of biannual [[alpha-fetoprotein]] and [[ultrasonography]], as compared to [[no screening]], for mortality from hepatocellular carcinoma was 0.6 and the [[relative risk reduction]] was 36.7%. In populations similar to those in this study which had a rate of risk as measured by the mortality from hepatocellular carcinoma of 0.1315% without treatment, the [[number needed to treat]] is 2070. <ref name="pmid15042359"/> | |||
==References== | |||
<references/> | |||
==References cited== | |||
{{Reflist3 test|refs= | |||
<ref name="pmid11350553">{{cite journal| author=Tong MJ, Blatt LM, Kao VW| title=Surveillance for hepatocellular carcinoma in patients with chronic viral hepatitis in the United States of America. | journal=J Gastroenterol Hepatol | year= 2001 | volume= 16 | issue= 5 | pages= 553-9 | pmid=11350553 | |||
| url=http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=clinical.uthscsa.edu/cite&email=badgett@uthscdsa.edu&retmode=ref&cmd=prlinks&id=11350553 }}</ref> | |||
<ref name="pmid15042359">{{cite journal| author=Zhang BH, Yang BH, Tang ZY| title=Randomized controlled trial of screening for hepatocellular carcinoma. | journal=J Cancer Res Clin Oncol | year= 2004 | volume= 130 | issue= 7 | pages= 417-22 | pmid=15042359 | doi=10.1007/s00432-004-0552-0 | pmc= | url= }} </ref> | |||
<ref name="pmid17767898">{{cite journal| author=Thompson Coon J, Rogers G, Hewson P, Wright D, Anderson R, Cramp M et al.| title=Surveillance of cirrhosis for hepatocellular carcinoma: systematic review and economic analysis. | journal=Health Technol Assess | year= 2007 | volume= 11 | issue= 34 | pages= 1-206 | pmid=17767898 | doi= | pmc= | url=http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=17767898 }} </ref> | |||
<ref name="pmid21374666">{{cite journal| author=Bruix J, Sherman M, American Association for the Study of Liver Diseases| title=Management of hepatocellular carcinoma: an update. | journal=Hepatology | year= 2011 | volume= 53 | issue= 3 | pages= 1020-2 | pmid=21374666 | doi=10.1002/hep.24199 | pmc=PMC3084991 | url= }} </ref> | |||
}} | |||
Revision as of 15:25, 19 October 2011
Diagnosis
The alpha-fetoprotein may be elevated; however, its positive predictive value is low and it may be elevated in patients with cirrhosis.[1]
Treatment
Hepatocellular cancer treatment information from the National Cancer Institute's Physician Data Query
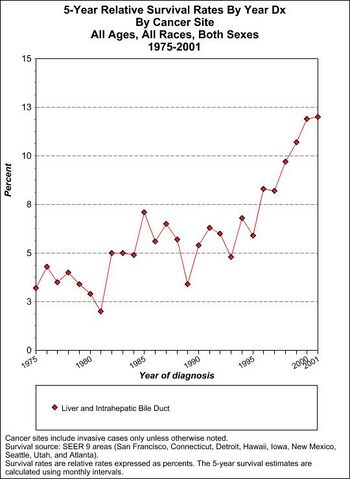
Prognosis
Staging information
Hepatocellular cancer staging information from the National Cancer Institute's Physician Data Query
Screening
"Surveillance is deemed cost-effective if the expected HCC risk exceeds 1.5% per year in patients with hepatitis C and 0.2% per year in patients with hepatitis B. Analysis of recent studies show that alpha-fetoprotein determination lacks adequate sensitivity and specificity for effective surveillance (and for diagnosis). Thus, surveillance has to be based on ultrasound examination. The recommended screening interval is 6 months" according to a clinical practice guideline by the American Association for the Study of Liver Diseases. [2] "In a mixed-aetiology cohort, the most effective surveillance strategy is to screen each patient with AFP assay and ultrasound imaging on a 6-monthly basis" according to a systematic review by the NIHR Health Technology Assessment programme (UK). [3] In a randomized controlled trial, the relative risk ratio of biannual alpha-fetoprotein and ultrasonography, as compared to no screening, for mortality from hepatocellular carcinoma was 0.6 and the relative risk reduction was 36.7%. In populations similar to those in this study which had a rate of risk as measured by the mortality from hepatocellular carcinoma of 0.1315% without treatment, the number needed to treat is 2070. [4]
References
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid11350553 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid21374666 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid17767898 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid15042359
References cited
Cite error: <ref> tag with name "pmid11350553" defined in <references> has group attribute "" which does not appear in prior text.
Cite error: <ref> tag with name "pmid15042359" defined in <references> has group attribute "" which does not appear in prior text.
Cite error: <ref> tag with name "pmid17767898" defined in <references> has group attribute "" which does not appear in prior text.
Cite error: <ref> tag with name "pmid21374666" defined in <references> has group attribute "" which does not appear in prior text.