User:Milton Beychok/Sandbox: Difference between revisions
imported>Milton Beychok mNo edit summary |
imported>Milton Beychok No edit summary |
||
| Line 1: | Line 1: | ||
The '''acid dew point''' (also '''acid dewpoint''') of a [[flue gas]] (i.e., a [[combustion]] product gas) is the [[temperature]], at a given [[pressure]], at which any gaseous [[acid]] in the flue gas will start to [[condensation|condense]] into liquid acid. The acid dew point of a flue gas, at a given pressure, is often referred to as the point at which the flue gas is "saturated" with gaseous acid, meaning that the flue gas cannot hold any more gaseous acid.<ref>{{cite book|author=David A. Lewandowski|title=Design of Thermal Oxidation Systems for Volatile Organic Compounds|edition=1st Edition|publisher=CRC Press|year=2000|id=ISBN 1-56670-410-3}}</ref><ref>{{cite book|author=Walter R. Niessen|title=Combustion and Incineration Processes|edition=3rd Edition|publisher=CRC Press|year=2002|id=ISBN 0-8247-0629-3}}</ref><ref>{{cite journal|author=W.M.M. Huijbregts and R. Leferink|title=Latest Advances in the Understanding of Acid Dewpoint Corrosion: Corrosion and Stress Corrosion Cracking in Combustion Gas Condensates|journal=Anti-Corrosion Methods and Materials|volume=51|issue=3|pages=173 - 188|date=2004|id=|url=http://www.hbscc.nl/pdf/56%20ACMM%20Condensate%20%20SCC.pdf}}</ref> | |||
==Chemistry and mechanism== | |||
===Sulfuric acid dewpoint== | |||
{{Image|Sulfuric acid dew point.png|right|397px|Calculated sulfuric acid dew points of typical combustion flue gases, as a function of SO3 content, and water vapor content <ref>[http://www.condexenergy.com/Condensing_Economizer_Article.pdf Condensing Economizer Article]</ref>}} | {{Image|Sulfuric acid dew point.png|right|397px|Calculated sulfuric acid dew points of typical combustion flue gases, as a function of SO3 content, and water vapor content <ref>[http://www.condexenergy.com/Condensing_Economizer_Article.pdf Condensing Economizer Article]</ref>}} | ||
As a broad generality, flue gases from the combustion of [[coal]], [[fuel oil]], [[natural gas]], or [[biomass]] are primarily composed of [[carbon dioxide]] (CO<sub>2</sub>) and [[water]] vapor (H<sub>2</sub>O) as well as [[nitrogen]] (N<sub>2</sub>) and excess [[oxygen]] (O<sub>2</sub>) remaining from the intake combustion [[air]]. Typically, more than two-thirds of the flue gas is nitrogen. The combustion flue gases may also contain small amounts of [[particulate matter]], [[carbon monoxide]], [[nitrogen oxide]]s, and [[sulfur oxide]]s in the form of gaseous [[sulfur dioxide]] (SO<sub>2</sub>) and gaseous [[sulfur trioxide]] (SO<sub>3</sub>). The SO<sub>3</sub> is present because a portion of the SO<sub>2</sub> formed in the combustion of the [[sulfur]] (S) compounds in the combustion fuel is further oxidized to SO<sub>3</sub>. The gas phase SO<sub>3</sub> then combines the vapor phase H<sub>2</sub>O to form gas phase sulfuric acid H<sub>2</sub>SO<sub>4</sub>: | |||
As a broad generality, | |||
:H<sub>2</sub>O + SO<sub>3</sub> → H<sub>2</sub>SO<sub>4</sub> | :: H<sub>2</sub>O + SO<sub>3</sub> → H<sub>2</sub>SO<sub>4</sub> | ||
:water + sulfur trioxide → sulfuric acid | |||
Because of the presence of | Because of the presence of gaseous sulfuric acid, the sulfuric acid dew point of most flue gases is much higher than the [[water dew point]] of the flue gases. For example, a flue gas with 5 volume % water vapor and containing no acid gases has a water dew point of about 32 °C (90 °F). The same flue gas with the addition of only 0.01 volume percent of SO<sub>3</sub> will have a sulfuric acid dew point of about 118 °C (244 °F).<ref>[http://www.banksengineering.com/Burning%20Sulfur%20Compounds.pdf Burning Sulfur Compounds] A publication of the Banks Engineering Company of [[Oklahoma]].</ref> | ||
The acid dew point of a combustion flue gas depends upon the composition of the specific fuel being burned and the resultant composition of the flue gas. Given a flue gas composition, its acid dew point can be predicted fairly closely. As an approximation, the acid dew points of flue gases from thermal power | The acid dew point of a combustion flue gas depends upon the composition of the specific fuel being burned and the resultant composition of the flue gas. Given a flue gas composition, its acid dew point can be predicted fairly closely. As an approximation, the sulfuric acid dew points of flue gases from [[thermal power plant]]s range from about 120 °C to about 150 °C (250 to 300 °F). | ||
== | ==Calculation of acid dewpoints== | ||
'''''Sulfuric acid (H<sub>2</sub>SO<sub>4</sub>) dew point:''''' <ref>{{cite journal|author=F.H. Verhoff and J.T. Banchero|title=Predicting Dew Points of Gases |journal=Chemical Engineering progress|volume=78|issue=8|pages=71 - 72|date=1974|id=|url=}}</ref><ref>{{cite journal|author=R.R. Pierce|title=Estimating Acid Dewpoints in Stack Gases|journal=Chemical Engineering|volume=84|issue=8|pages=125 - 128|date=1977|id=|url=}}</ref> | '''''Sulfuric acid (H<sub>2</sub>SO<sub>4</sub>) dew point:''''' <ref>{{cite journal|author=F.H. Verhoff and J.T. Banchero|title=Predicting Dew Points of Gases |journal=Chemical Engineering progress|volume=78|issue=8|pages=71 - 72|date=1974|id=|url=}}</ref><ref>{{cite journal|author=R.R. Pierce|title=Estimating Acid Dewpoints in Stack Gases|journal=Chemical Engineering|volume=84|issue=8|pages=125 - 128|date=1977|id=|url=}}</ref> | ||
Revision as of 22:16, 21 May 2010
The acid dew point (also acid dewpoint) of a flue gas (i.e., a combustion product gas) is the temperature, at a given pressure, at which any gaseous acid in the flue gas will start to condense into liquid acid. The acid dew point of a flue gas, at a given pressure, is often referred to as the point at which the flue gas is "saturated" with gaseous acid, meaning that the flue gas cannot hold any more gaseous acid.[1][2][3]
Chemistry and mechanism
=Sulfuric acid dewpoint
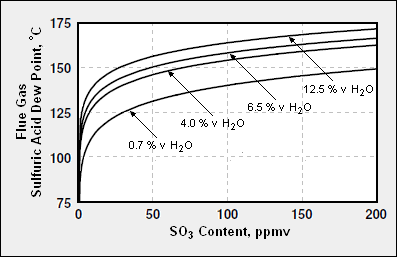
Calculated sulfuric acid dew points of typical combustion flue gases, as a function of SO3 content, and water vapor content [4]
As a broad generality, flue gases from the combustion of coal, fuel oil, natural gas, or biomass are primarily composed of carbon dioxide (CO2) and water vapor (H2O) as well as nitrogen (N2) and excess oxygen (O2) remaining from the intake combustion air. Typically, more than two-thirds of the flue gas is nitrogen. The combustion flue gases may also contain small amounts of particulate matter, carbon monoxide, nitrogen oxides, and sulfur oxides in the form of gaseous sulfur dioxide (SO2) and gaseous sulfur trioxide (SO3). The SO3 is present because a portion of the SO2 formed in the combustion of the sulfur (S) compounds in the combustion fuel is further oxidized to SO3. The gas phase SO3 then combines the vapor phase H2O to form gas phase sulfuric acid H2SO4:
- H2O + SO3 → H2SO4
- water + sulfur trioxide → sulfuric acid
Because of the presence of gaseous sulfuric acid, the sulfuric acid dew point of most flue gases is much higher than the water dew point of the flue gases. For example, a flue gas with 5 volume % water vapor and containing no acid gases has a water dew point of about 32 °C (90 °F). The same flue gas with the addition of only 0.01 volume percent of SO3 will have a sulfuric acid dew point of about 118 °C (244 °F).[5]
The acid dew point of a combustion flue gas depends upon the composition of the specific fuel being burned and the resultant composition of the flue gas. Given a flue gas composition, its acid dew point can be predicted fairly closely. As an approximation, the sulfuric acid dew points of flue gases from thermal power plants range from about 120 °C to about 150 °C (250 to 300 °F).
Calculation of acid dewpoints
Sulfuric acid (H2SO4) dew point: [6][7]
(1)
or this equivalent form:
(2)
Sulfurous acid (H2SO3) dew point: [8]
(3)
Hydrochloric acid (HCl) dewpoint: [8]
(4)
Nitric acid (HNO3) dewpoint: [8]
(4)
References
- ↑ David A. Lewandowski (2000). Design of Thermal Oxidation Systems for Volatile Organic Compounds, 1st Edition. CRC Press. ISBN 1-56670-410-3.
- ↑ Walter R. Niessen (2002). Combustion and Incineration Processes, 3rd Edition. CRC Press. ISBN 0-8247-0629-3.
- ↑ W.M.M. Huijbregts and R. Leferink (2004). "Latest Advances in the Understanding of Acid Dewpoint Corrosion: Corrosion and Stress Corrosion Cracking in Combustion Gas Condensates". Anti-Corrosion Methods and Materials 51 (3): 173 - 188.
- ↑ Condensing Economizer Article
- ↑ Burning Sulfur Compounds A publication of the Banks Engineering Company of Oklahoma.
- ↑ F.H. Verhoff and J.T. Banchero (1974). "Predicting Dew Points of Gases". Chemical Engineering progress 78 (8): 71 - 72.
- ↑ R.R. Pierce (1977). "Estimating Acid Dewpoints in Stack Gases". Chemical Engineering 84 (8): 125 - 128.
- ↑ 8.0 8.1 8.2 Yen Hsiung Kiang (1981). "Predicting Dewpoints of Gases". Chemical Engineering 88 (3): 127.




