User:Milton Beychok/Sandbox: Difference between revisions
Jump to navigation
Jump to search
imported>Milton Beychok No edit summary |
imported>Milton Beychok No edit summary |
||
| Line 1: | Line 1: | ||
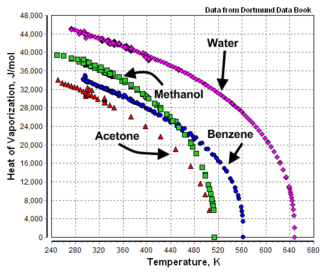
{{Image|Heat of Vaporization.png|right|325px|Heats of vaporization versus temperature.}} | {{Image|Heat of Vaporization.png|right|325px|Heats of vaporization versus temperature.<ref>[http://www.ddbst.com/new/Default.htm Dortmund Data Bank Online Search]</ref>}} | ||
{| class = "wikitable" align=" | <br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br> | ||
{| class = "wikitable" align="right" | |||
|+ Heat of vaporization, normal boiling point and critical<br/>temperature and pressure of various liquids <ref>{{cite book|author=J.M. Smith, H.C. Van Ness and N.M. Abbot|title=Introduction to Chemical Engineering Thermodynamics|edition=7th Edition|publisher=McGraw-Hill|year=2004|id=ISBN 0-07-310445-0}}</ref><ref>{{cite book|author=Robert C. Weast (Editor)|title=Perry's Chemical Engineers' Handbook|edition=56th Edition|publisher=CRC Press|year=1976|id=ISBN 0-87819-455-X}}</ref> | |+ Heat of vaporization, normal boiling point and critical<br/>temperature and pressure of various liquids <ref>{{cite book|author=J.M. Smith, H.C. Van Ness and N.M. Abbot|title=Introduction to Chemical Engineering Thermodynamics|edition=7th Edition|publisher=McGraw-Hill|year=2004|id=ISBN 0-07-310445-0}}</ref><ref>{{cite book|author=Robert C. Weast (Editor)|title=Perry's Chemical Engineers' Handbook|edition=56th Edition|publisher=CRC Press|year=1976|id=ISBN 0-87819-455-X}}</ref> | ||
|- | |- | ||
| Line 13: | Line 15: | ||
!colspan=2|( atm ) | !colspan=2|( atm ) | ||
|- align="center" | |- align="center" | ||
|Acetic acid||C<sub>2</sub>H<sub>4</sub>O<sub>2</sub>||colspan=2|23,700||117.9||391.1||colspan=2|594.8||colspan=2|57.1 | |[[Acetic acid]]||C<sub>2</sub>H<sub>4</sub>O<sub>2</sub>||colspan=2|23,700||117.9||391.1||colspan=2|594.8 | ||
||colspan=2|57.1 | |||
|- align="center" | |- align="center" | ||
| Acetone||C<sub>3</sub>H<sub>6</sub>O||colspan=2|29,100||56.2||329.4||colspan=2|508.7||colspan=2|47.0 | | [[Acetone]]||C<sub>3</sub>H<sub>6</sub>O||colspan=2|29,100||56.2||329.4||colspan=2|508.7||colspan=2|47.0 | ||
|- align="center" | |- align="center" | ||
|Benzene||C<sub>6</sub>H<sub>6</sub>||colspan=2|30,720||80.0||353.2||colspan=2|562.1||colspan=2|48.6 | |[[Benzene]]||C<sub>6</sub>H<sub>6</sub>||colspan=2|30,720||80.0||353.2||colspan=2|562.1||colspan=2|48.6 | ||
|- align="center" | |- align="center" | ||
| | |[[Butane]]||C<sub>4</sub>H<sub>10</sub>||colspan=2|22,440||– 0.5||272.7||colspan=2|425.2||colspan=2|37.5 | ||
|- align="center" | |- align="center" | ||
| | |[[Carbon tetrachloride]]||CCl<sub>4</sub>||colspan=2|29,820||76.6||349.8||colspan=2|556.3||colspan=2|45.0 | ||
|- align="center" | |- align="center" | ||
| | |[[Chloroform]]||CHCl<sub>3</sub>||colspan=2|29,240||61.1||334.3||colspan=2|536.2||colspan=2|54.0 | ||
|- align="center" | |- align="center" | ||
| | |[[Cyclohexane]]||C<sub>6</sub>H<sub>12</sub>||colspan=2|29,970||80.7||353.9||colspan=2|553.6||colspan=2|40.0 | ||
|- align="center" | |- align="center" | ||
| | |[[Cyclopentane]]||C<sub>5</sub>H<sub>10</sub>||colspan=2|27,300||49.2||322.4||colspan=2|511.8||colspan=2|44.6 | ||
|- align="center" | |- align="center" | ||
| | |[[Decane]]||C<sub>10</sub>H<sub>22</sub>||colspan=2|38,750||174.1||447.3||colspan=2|617.6 ||colspan=2|20.8 | ||
|- align="center" | |- align="center" | ||
| | |[[Ethanol]]||C<sub>2</sub>H<sub>6</sub>O||colspan=2|38,560||78.2||351.4||colspan=2|516.2||colspan=2|63.0 | ||
|- align="center" | |- align="center" | ||
| | |[[Hexane]]||C<sub>6</sub>H<sub>14</sub>||colspan=2|28.850||68.7||341.9||colspan=2|507.4||colspan=2|29.9 | ||
|- align="center" | |- align="center" | ||
|- align="center" | |- align="center" | ||
|Methanol||CH<sub>4</sub>O||colspan=2|35,210||64.7||337.9||colspan=2|513.2||colspan=2|78.5 | |[[Isobutane]]||C<sub>4</sub>H<sub>10</sub>||colspan=2|21,300||– 11.9||261.3||colspan=2|408.2||colspan=2|36.0 | ||
|- | |||
|[[Methanol]]||CH<sub>4</sub>O||colspan=2|35,210||64.7||337.9||colspan=2|513.2||colspan=2|78.5 | |||
|- align="center" | |- align="center" | ||
| | |[[Octane]]||C<sub>8</sub>H<sub>18</sub>||colspan=2|34,410||125.6||398.8||colspan=2|569.2||colspan=2|24.8 | ||
|- align="center" | |- align="center" | ||
|Water||H<sub>2</sub>0||colspan=2|40,660||100||373.2||colspan=2|647.3||colspan=2|218.3 | |[[Water]]||H<sub>2</sub>0||colspan=2|40,660||100||373.2||colspan=2|647.3||colspan=2|218.3 | ||
|} | |} | ||
<br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br> | |||
<br><br><br><br><br><br><br><br><br><br><br><br> | |||
==References== | ==References== | ||
{{reflist}} | {{reflist}} | ||
Revision as of 00:29, 8 September 2009
| Name | Formula | Hv | Tn | Tc | Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| ( J/mol ) | ( °C ) | ( K ) | ( K ) | ( atm ) | |||||
| Acetic acid | C2H4O2 | 23,700 | 117.9 | 391.1 | 594.8 | colspan=2|57.1 | |||
| Acetone | C3H6O | 29,100 | 56.2 | 329.4 | 508.7 | 47.0 | |||
| Benzene | C6H6 | 30,720 | 80.0 | 353.2 | 562.1 | 48.6 | |||
| Butane | C4H10 | 22,440 | – 0.5 | 272.7 | 425.2 | 37.5 | |||
| Carbon tetrachloride | CCl4 | 29,820 | 76.6 | 349.8 | 556.3 | 45.0 | |||
| Chloroform | CHCl3 | 29,240 | 61.1 | 334.3 | 536.2 | 54.0 | |||
| Cyclohexane | C6H12 | 29,970 | 80.7 | 353.9 | 553.6 | 40.0 | |||
| Cyclopentane | C5H10 | 27,300 | 49.2 | 322.4 | 511.8 | 44.6 | |||
| Decane | C10H22 | 38,750 | 174.1 | 447.3 | 617.6 | 20.8 | |||
| Ethanol | C2H6O | 38,560 | 78.2 | 351.4 | 516.2 | 63.0 | |||
| Hexane | C6H14 | 28.850 | 68.7 | 341.9 | 507.4 | 29.9 | |||
| Isobutane | C4H10 | 21,300 | – 11.9 | 261.3 | 408.2 | 36.0 | |||
| Methanol | CH4O | 35,210 | 64.7 | 337.9 | 513.2 | 78.5 | |||
| Octane | C8H18 | 34,410 | 125.6 | 398.8 | 569.2 | 24.8 | |||
| Water | H20 | 40,660 | 100 | 373.2 | 647.3 | 218.3 | |||
References
- ↑ Dortmund Data Bank Online Search
- ↑ J.M. Smith, H.C. Van Ness and N.M. Abbot (2004). Introduction to Chemical Engineering Thermodynamics, 7th Edition. McGraw-Hill. ISBN 0-07-310445-0.
- ↑ Robert C. Weast (Editor) (1976). Perry's Chemical Engineers' Handbook, 56th Edition. CRC Press. ISBN 0-87819-455-X.