User:Milton Beychok/Sandbox: Difference between revisions
imported>Milton Beychok |
imported>Milton Beychok |
||
| Line 16: | Line 16: | ||
Raw natural gas consists primarily of methane (CH<sub>4</sub>), the shortest [[hydrocarbon]] molecule. It also contains various amounts of heavier hydrocarbon gases such as [[ethane]] (C<sub>2</sub>H<sub>6</sub>), [[propane]] (C<sub>3</sub>H<sub>8</sub>), [[Butane|normal butane]] (n-C<sub>4</sub>H<sub>10</sub>), [[isobutane]] (i-C<sub>4</sub>H<sub>10</sub>), [[pentane]]s and even higher [[molecular weight]] hydrocarbons. The raw gas also contains various amounts of [[acid gas]]es such as [[carbon dioxide]] (CO<sub>2</sub>), [[hydrogen sulfide]] (H<sub>2</sub>S) and [[mercaptan]]s such as [[methanethiol]] (CH<sub>3</sub>SH) and [[ethanethiol]] {C<sub>2</sub>H<sub>5</sub>SH). | Raw natural gas consists primarily of methane (CH<sub>4</sub>), the shortest [[hydrocarbon]] molecule. It also contains various amounts of heavier hydrocarbon gases such as [[ethane]] (C<sub>2</sub>H<sub>6</sub>), [[propane]] (C<sub>3</sub>H<sub>8</sub>), [[Butane|normal butane]] (n-C<sub>4</sub>H<sub>10</sub>), [[isobutane]] (i-C<sub>4</sub>H<sub>10</sub>), [[pentane]]s and even higher [[molecular weight]] hydrocarbons. The raw gas also contains various amounts of [[acid gas]]es such as [[carbon dioxide]] (CO<sub>2</sub>), [[hydrogen sulfide]] (H<sub>2</sub>S) and [[mercaptan]]s such as [[methanethiol]] (CH<sub>3</sub>SH) and [[ethanethiol]] {C<sub>2</sub>H<sub>5</sub>SH). | ||
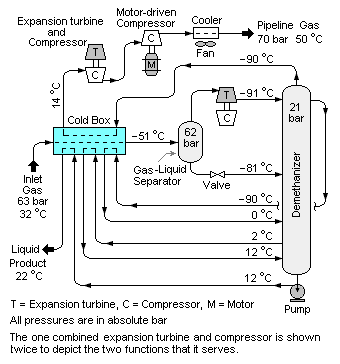
When processed into finished by-products (see [[Natural gas processing]]), these heavier hydrocarbons are collectively referred to as NGL (natural gas liquids). The extraction and recovery of the NGl often involves using an expansion turbine and a low-temperature distillation column ( | When processed into finished by-products (see [[Natural gas processing]]), these heavier hydrocarbons are collectively referred to as NGL (natural gas liquids). The extraction and recovery of the NGl often involves using an expansion turbine and a low-temperature distillation column (commonly referred to as a demethanizer) as shown in Figure 1. | ||
Revision as of 13:57, 10 July 2008
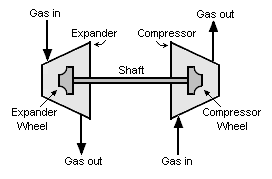
An expansion turbine, also referred to as a turboexpander or turbo-expander, is a centrifugal or axial flow turbine through which a high pressure gas is expanded to produce work that is often used to drive a gas compressor.
Because work is extracted from the expanding high pressure gas, the expansion is an isentropic process (i.e., a constant entropy process) and the low pressure exhaust gas from the turbine is at a very low temperature, sometimes as low as -90 °C or less.
Expansion turbines are very widely used as sources of refrigeration in industrial processes such as the extraction of ethane and natural gas liquids (NGLs) from natural gas,[1] the liquefaction of gases (such as oxygen, nitrogen, helium, argon and krypton)[2][3] and other low-temperature processes.
Applications
Although expansion turbines are very commonly used in low-temperature processes, they are used in many other applications as well. This section discusses one of the low temperature procesees as well as some of the other applications.
Extracting hydrocarbon liquids from natural gas
Raw natural gas consists primarily of methane (CH4), the shortest hydrocarbon molecule. It also contains various amounts of heavier hydrocarbon gases such as ethane (C2H6), propane (C3H8), normal butane (n-C4H10), isobutane (i-C4H10), pentanes and even higher molecular weight hydrocarbons. The raw gas also contains various amounts of acid gases such as carbon dioxide (CO2), hydrogen sulfide (H2S) and mercaptans such as methanethiol (CH3SH) and ethanethiol {C2H5SH).
When processed into finished by-products (see Natural gas processing), these heavier hydrocarbons are collectively referred to as NGL (natural gas liquids). The extraction and recovery of the NGl often involves using an expansion turbine and a low-temperature distillation column (commonly referred to as a demethanizer) as shown in Figure 1.
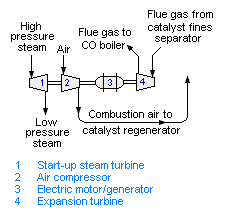
Power recovery in fluid catalytic cracker
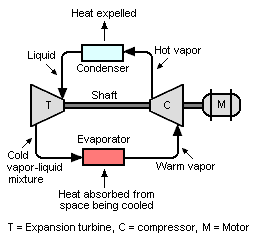
Refrigeration system
Power generation
History
In 1939, Pyotr Kapitza of Russia suggested the use of a centrifugal turbine for the isentropic expansion of gases to produce refrigeration. Since then, centrifugal expansion turbines have taken over almost 100 percent of the gas liquefaction and other low-temperature industrial requirements.
References
- ↑ Demethanzer
- ↑ BOC (NZ) publication: use search function for keyword "expansion"
- ↑ US Department of Energy Hydrogen Program