Human papilloma virus (HPV): Difference between revisions
imported>Aryeh Sokolov |
imported>Aryeh Sokolov |
||
| Line 21: | Line 21: | ||
== Natural Host: == | == Natural Host: == | ||
HPV | The only host for HPV are Humans. | ||
== Discovery and Link to Cancer == | == Discovery and Link to Cancer == | ||
Revision as of 01:31, 13 May 2009
For the course duration, the article is closed to outside editing. Of course you can always leave comments on the discussion page. The anticipated date of course completion is May 21, 2009. One month after that date at the latest, this notice shall be removed. Besides, many other Citizendium articles welcome your collaboration! |
| Human papilloma virus | ||||||
|---|---|---|---|---|---|---|
| Virus classification | ||||||
| ||||||
| Vectors | ||||||
|
human |
Description and significance
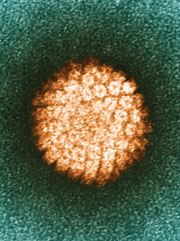
Papillomaviruses are nonenveloped DNA viruses. These viruses are diverse, but all can infect the skin and mucosal tissues of a many vertebrate species, including humans.[1] A group of genital mucosotropic human papilloma virus (HPV) types are etiologic agents responsible for virtually all cases of cervical cancer, as well as a substantial fraction of other ano-genital and head-and-neck cancers (reviewed in [1]). Cancer-associated genital HPV types, as well as another subset of HPV types associated with the development of benign genital warts (condyloma accuminata), are generally transmitted through sexual contact. Infection with genital HPV types is very common, with an estimated lifetime risk of infection of about 75% [2]. Although most genital HPV infections are subclinical and self-limiting, a subset of persistently infected individuals have lesions that progress to premalignancy or cancer.
Genome structure
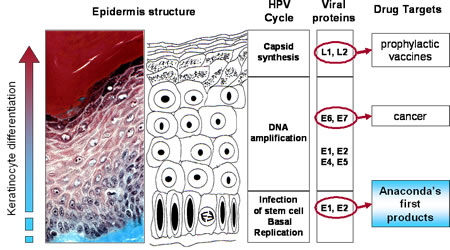
The HPV genome is a circular, double stranded DNA virus of approximately 8,000 base pairs and made up of 40-50% Guanine and Cytosine base pairs. All Papillomavirus genes are coded in one of the two DNA strands, using three forward reading frames, and alternative splicing for the individual expression of each gene. Papillomaviruses have the same general genomic organization. It consists of 8 open reading frames(ORF) all located on the same strand. These ORFs are designated either early (E) or late(L). The early ORFs-E1,E2,E4,E5,E6,and E7-code for nonstructural regulatory proteins. The E3 ORF does not code for a protein, and the E8 ORF has been identified only in bovine Papillomaviruses. The late ORFs-L1 and L2-code for capsid proteins. A 1-Kb non-coding region known as the upstream regulatory region (URR) ,lies between the early and late ORFs. The URR includes the origin of replication (ori), the E6/E7 gene promoter, and enhancers and silencers.
Natural Host:
The only host for HPV are Humans.
Discovery and Link to Cancer
In the early 1980s German Virologist Harold Zur Hausen discovered the link between cervical cancer and HPV. Using Southern Blot Hybridization, he went against the prevailing notion at the time that herpes simplex virus caused cervical cancer and discovered in fact that HPV DNA was present in the cervical tumors he tested. He named the first strain he discovered HPV-16. When another strain was discovered in about half the tumors he tested he named it HPV-18. His groundbreaking discovery led to the development of a vaccine and he was recognized with the 2008 Nobel Prize for physiology or medicine.
Interesting Features:
It is extremely difficult to grow HPV in tissue culture and laboratory animals. Currently, this has not been done successfully. However, techniques such as polymerase chain reaction (PCR) have are used to differentiate between different types. A distinct HPV type is defined as having less than 90% DNA base-pair homology with another identified HPV type.
How does this organism cause disease?
HPV can infect the stratified sq. epithelium and nonkeratinized epithelium (i.e mouth, upper airway, vagina, cervix, and anal canal). HPV can also induce tumors in other tissues such as the conjunctiva, lachrymal sac, nasal passages, bronchi, esophagus cervical glandular tissue, and the bladder. The cellular receptors suggested in virus binding including a6 Integrin and heparin sulfate glycosomaminoglycans, have been identified on keratinocytes. HPV can also be found near sites of infection as well. The vast majority of HPV infections are latent(asymptomatic).
Cancer and latency
HPV-induced malignant transformation appears to be the result of a complex series of events that are independent of viral reproduction. In latency, histopathologic changes are absent and no viral particles are produced. Although the vast majority of HPV infections are latent, the viral and cellular factors that abrogate, induce and maintain latency are unknown. Integration of HPV DNA into the host genome seems to be associated with the progression of neoplasia to cancer. Possible sites of viral integration in the host genome are numerous and may exist on a variety of chromosomes and in proximity to cellular oncogenes. Integration results in the disruption, deletion or inactivation of the E2 ORF. Integration may also disrupt other viral genes but E6, 7 are usually spared.
Current Research:
Epidemiology
23% of women attending sexually transmitted disease, family planning, and primary care outpatient clinics in the United States may be positive for high-risk HPV.[2] Geographic distribution of HPV is limited. Geographic disparities in the prevalence of common warts can be seen within countries. A worldwide prevalence survey of HPV types showed that HPV-16 is the most prevalent type(50%) overall, followed by HPV-18(14%), HPV-45(8%) and HPV-31(5%). But notable geographic locations exist, particularly among less common HPV types. HPV infections are endemic. No seasonality is recognized generally in the acquisition of HPV infection or the expression of HPV diseases. An increase of planar warts has been noted in the winter moths. The Prevalence of cervical HPV in women ranges from about 5-45% in Western Countries. These rates peak in young adults by their 3rd decade. Multiple HPV infections occur in 20-30% of HPV infected women. Positivity tests are usually transient. 13% of asymptomatic men tested positive for HPV DNA in one study. Cutaneous warts are very common among the population. . Hand warts represent 70% of all cutaneous warts and are mostly a childhood disease. The prevalence of common warts ranges from 0.8-22%. Plantar warts compose up to a third of cutaneous warts and are found in a wider age group, primarily in adolescents and young adults. Flat or juvenile warts constitute 4-8% of cutaneous warts with a peak prevalence among 10-12 yr olds. Genital HPV disease: the incidence is estimated to be ~5.5million new cases a year in the US, making it the most commonly acquired sexually transmitted disease. At least 20 million people are believed to be actively infected in the US, and it is estimated that ¾ of the population have been infected at one time. The prevalence of condyloma acuminatum (anal warts) is estimated to be 1% in the general pop. The incidence of anogenital warts is increasing. The number of initial visits for genital warts has increased fivefold between 1966 and 1987. HPV infection causes a vast majority of cervical cancer. HPV 16 and 18 are responsible for 70% of cases. Incidence increases directly with number of sexual partners. Low prevalence in nuns. Susceptibility increase if a male consort had partner with cervical cancer. Recurrent respiratory papillomatosis: The age of onset has a bimodal distribution which includes young adults an young children but not the elderly. Mainly associated with HPV6, 11. Affects the upper respiratory tract and median age is ~3years. With the juvenile onset form, the children are often delivered vaginally by teenage mothers with anogenital warts. For the adult-onset form, patients have a large number of lifetime sexual partners and a high frequency of oral sex. Risk Factors: cutaneous warts: use of heated swimming pools or of communal baths is activities that appear to promote the acquisition of plantar warts. People who work with meat poultry are, or fish are uniquely prone to had warts; up to 50% are infected. Unusual because predominant strain is HPV-7. Epidermodysplasia Verruciformis: susceptibility to EV appears to be governed by heterogeneous, non allelic genetic loci transmitted on an autosomal recessive or, more rarely, X-linked pattern. Anogenital warts: about 2/3 of the sexual partners of persons with anogenital warts will develop the disease within 2 years. The risk of developing genital warts is most strongly and directly linked to the lifetime number of sexual partners.
Prevention
I vitro studies show that carrageenan, extracted from red algae and commercially used to thicken products including sexual lubricants and infant formulas, is a potent inhibitor of HPV infection.[3]
HPV Vaccine
The HPV vaccine can reduce the incidence of high-grade cervical intraepithelial neoplasia[4] and HPV-associated anogenital disease[5].
The number needed to vaccinate to prevent the following diseases is estimated to be:[6]
- Condylomata acuminata (genital warts) is 8
- Cervical cancer is 324
A cost-benefit analysis concluded that the cost is $43,600 per quality-adjusted life-year (QALY) gained.[6]
In the United States, the Centers for Disease Control and Prevention recommends vaccination of females starting at aged 11 - 12 years.[7]
References
- ↑ Index of Viruses - Papillomaviruses (2006). In: ICTVdB - The Universal Virus Database, version 4. Büchen-Osmond, C (Ed), Columbia University, New York, USA. http://www.ncbi.nlm.nih.gov/ICTVdb/Ictv/fs_index.htm
- ↑ Datta, S. D., Koutsky, L. A., Ratelle, S., Unger, E. R., Shlay, J., McClain, T., et al. (2008). Human Papillomavirus Infection and Cervical Cytology in Women Screened for Cervical Cancer in the United States, 2003-2005. Ann Intern Med, 148(7), 493-500.
- ↑ Buck CB, Thompson CD, Roberts JN, Müller M, Lowy DR, Schiller JT (2006). "Carrageenan is a potent inhibitor of papillomavirus infection". PLoS Pathog. 2 (7): e69. DOI:10.1371/journal.ppat.0020069. PMID 16839203. Research Blogging.
- ↑ The FUTURE II Study Group (May 2007). "Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions". N. Engl. J. Med. 356 (19): 1915–27. DOI:10.1056/NEJMoa061741. PMID 17494925. Research Blogging. ACP Journal Club review JournalWatch review
- ↑ Garland SM, Hernandez-Avila M, Wheeler CM, et al for the Females United to Unilaterally Reduce Endo/Ectocervical Disease (FUTURE) I Investigators (May 2007). "Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases". N. Engl. J. Med. 356 (19): 1928–43. DOI:10.1056/NEJMoa061760. PMID 17494926. Research Blogging. ACP Journal Club review JournalWatch review
- ↑ 6.0 6.1 Brisson M, Van de Velde N, De Wals P, Boily MC (August 2007). "Estimating the number needed to vaccinate to prevent diseases and death related to human papillomavirus infection". CMAJ 177 (5): 464–8. DOI:10.1503/cmaj.061709. PMID 17709404. PMC 1950193. Research Blogging.
Cite error: Invalid
<ref>tag; name "pmid17709404" defined multiple times with different content - ↑ Advisory Committee on Immunization Practices (ACIP) (March 12, 2007). Quadrivalent Human Papillomavirus Vaccine Recommendations of the Advisory Committee on Immunization Practices (ACIP). Centers for Disease Control and Prevention. Retrieved on 2008-11-25.