Darusentan: Difference between revisions
Jump to navigation
Jump to search

imported>David E. Volk No edit summary |
imported>David E. Volk mNo edit summary |
||
| (One intermediate revision by the same user not shown) | |||
| Line 1: | Line 1: | ||
{{subpages}} | |||
{{Chem infobox | {{Chem infobox | ||
|align=right | |align=right | ||
|image=[[Image:Darusentan.png|center|thumb| | |image=[[Image:Darusentan.png|center|thumb|300px]] | ||
|width= | |width=300px | ||
|molname=darusentan | |molname= darusentan | ||
|synonyms= | |synonyms= | ||
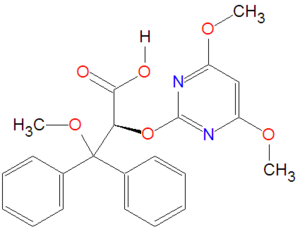
|molformula= C<sub>22</sub>H<sub>22</sub>N<sub>2</sub>O<sub>6</sub> | |molformula= C<sub>22</sub>H<sub>22</sub>N<sub>2</sub>O<sub>6</sub> | ||
Latest revision as of 08:59, 19 September 2009
|
| |||||||
| darusentan | |||||||
| |||||||
| Uses: | hypertension | ||||||
| Properties: | |||||||
| Hazards: | see side effects & drug interactions | ||||||
| |||||||
In medicine, darusentan is a medication for treating hypertension that is a selective endothelin type A antagonist. It is a vasodilator.[1]
Drug toxicity
Fluid retention is the most common drug toxicity.[1]
References
- ↑ 1.0 1.1 Weber MA et al. (2009) A selective endothelin-receptor antagonist to reduce blood pressure in patients with treatment-resistant hypertension: a randomised, double-blind, placebo-controlled trial. Lancet DOI:10.1016/S0140-6736(09)61500-2