User:Milton Beychok/Sandbox: Difference between revisions
imported>Milton Beychok mNo edit summary |
imported>Milton Beychok No edit summary |
||
| Line 15: | Line 15: | ||
As can be seen, the heat of vaporization of a liquid at a given temperature (other than the normal boiling point temperature) may vary significantly from the value reported at the normal boiling point of the liquid. | As can be seen, the heat of vaporization of a liquid at a given temperature (other than the normal boiling point temperature) may vary significantly from the value reported at the normal boiling point of the liquid. | ||
{| class = "wikitable" align="right" | {| class = "wikitable" align="right" | ||
| Line 69: | Line 66: | ||
|} | |} | ||
== Estimating heat of vaporization values == | |||
<br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br> | |||
==References== | ==References== | ||
{{reflist}} | {{reflist}} | ||
Revision as of 17:28, 8 September 2009
The heat of vaporization, () is the amount of thermal energy required to convert a quantity of liquid into a vapor. It can be thought of as the energy required to break the intermolecular bonds within the liquid.
It is also often referred to as the latent heat of vaporization ( or ) and the enthalpy of vaporization (symbol or ) and is usually measured and reported at the temperature corresponding to the normal boiling point of the liquid.
Measurement units
Heat of vaporization values are usually reported in measurement units such as J/mol or kJ/mol and referred to as the molar heat of vaporization, although J/g or kJ/kg are also often used. Older units such as kcal/mol, cal/g, Btu/lb and others are still used sometimes.
Temperature dependency
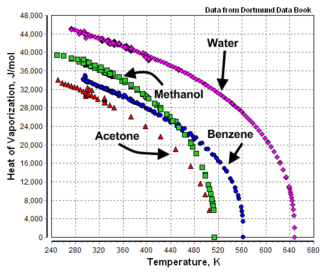
Heats of vaporization are temperature dependent as shown in Figure 1 by the example graphs of temperature versus heat of vaporization for acetone, benzene, methanol and water.
As can be seen, the heat of vaporization of a liquid at a given temperature (other than the normal boiling point temperature) may vary significantly from the value reported at the normal boiling point of the liquid.
| Name | Formula | Hv | Tn | Tc | Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| ( J/mol ) | ( °C ) | ( K ) | ( K ) | ( atm ) | |||||
| Acetic acid | C2H4O2 | 23,700 | 117.9 | 391.1 | 594.8 | 57.1 | |||
| Acetone | C3H6O | 29,100 | 56.2 | 329.4 | 508.7 | 47.0 | |||
| Benzene | C6H6 | 30,720 | 80.0 | 353.2 | 562.1 | 48.6 | |||
| Butane | C4H10 | 22,440 | – 0.5 | 272.7 | 425.2 | 37.5 | |||
| Carbon tetrachloride | CCl4 | 29,820 | 76.6 | 349.8 | 556.3 | 45.0 | |||
| Chloroform | CHCl3 | 29,240 | 61.1 | 334.3 | 536.2 | 54.0 | |||
| Cyclohexane | C6H12 | 29,970 | 80.7 | 353.9 | 553.6 | 40.0 | |||
| Cyclopentane | C5H10 | 27,300 | 49.2 | 322.4 | 511.8 | 44.6 | |||
| Decane | C10H22 | 38,750 | 174.1 | 447.3 | 617.6 | 20.8 | |||
| Ethanol | C2H6O | 38,560 | 78.2 | 351.4 | 516.2 | 63.0 | |||
| Hexane | C6H14 | 28.850 | 68.7 | 341.9 | 507.4 | 29.9 | |||
| Isobutane | C4H10 | 21,300 | – 11.9 | 261.3 | 408.2 | 36.0 | |||
| Methanol | CH4O | 35,210 | 64.7 | 337.9 | 513.2 | 78.5 | |||
| Octane | C8H18 | 34,410 | 125.6 | 398.8 | 569.2 | 24.8 | |||
| Water | H20 | 40,660 | 100 | 373.2 | 647.3 | 218.3 | |||
Notes:
| |||||||||
Estimating heat of vaporization values
References
- ↑ Dortmund Data Bank Online Search
- ↑ J.M. Smith, H.C. Van Ness and N.M. Abbot (2004). Introduction to Chemical Engineering Thermodynamics, 7th Edition. McGraw-Hill. ISBN 0-07-310445-0.
- ↑ Robert C. Weast (Editor) (1976). Perry's Chemical Engineers' Handbook, 56th Edition. CRC Press. ISBN 0-87819-455-X.